The solubility of a gas in a liquid increases with increasing pressure. To understand the above...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
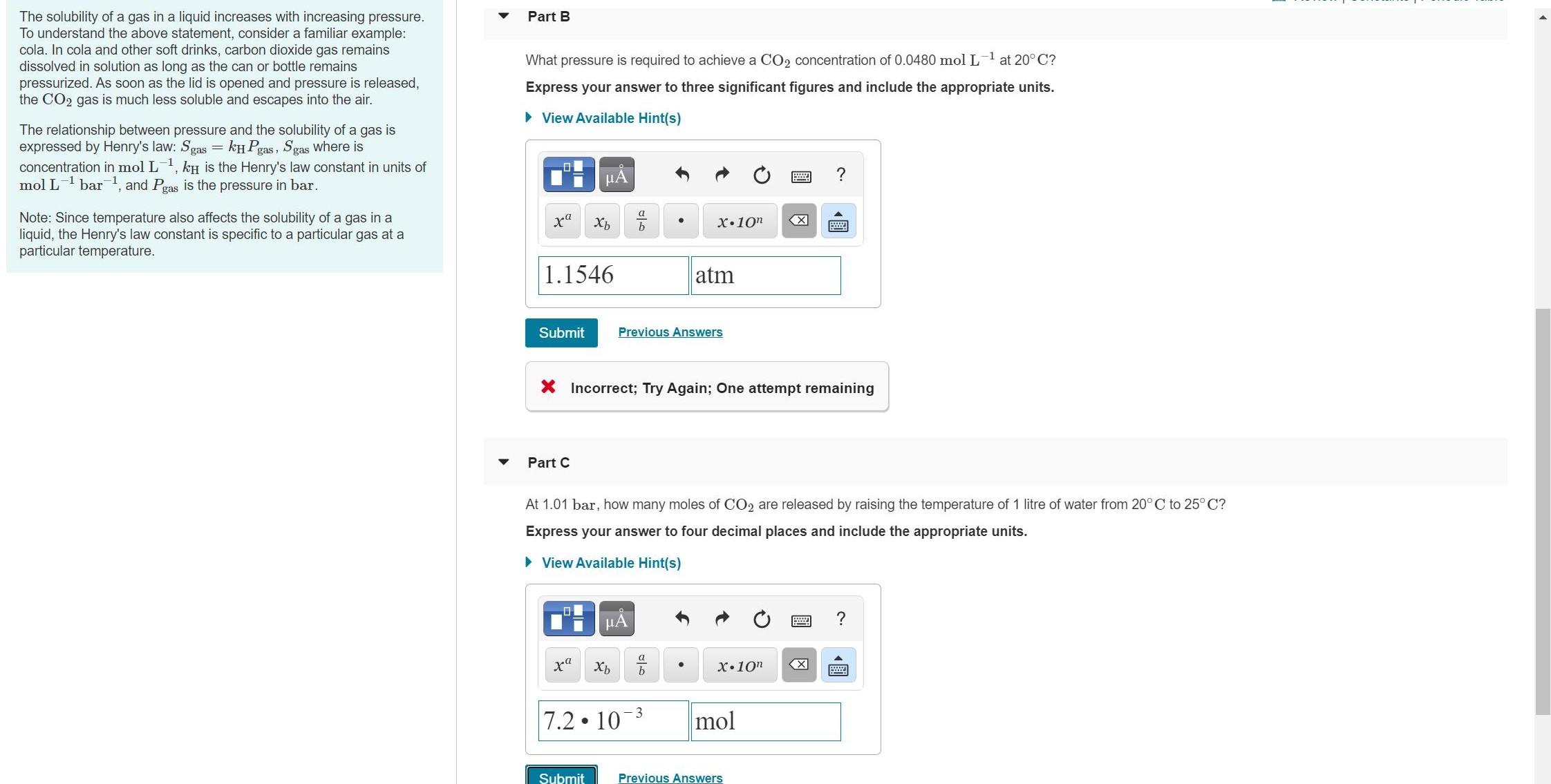
The solubility of a gas in a liquid increases with increasing pressure. To understand the above statement, consider a familiar example: cola. In cola and other soft drinks, carbon dioxide gas remains dissolved in solution as long as the can or bottle remains pressurized. As soon as the lid is opened and pressure is released, the CO2 gas is much less soluble and escapes into the air. Part B What pressure is required to achieve a CO2 concentration of 0.0480 mol L-1 at 20° C? Express your answer to three significant figures and include the appropriate units. • View Available Hint(s) The relationship between pressure and the solubility of a gas is expressed by Henry's law: Sgas = kH Pgas, Sgas Where is concentration in mol L, kH is the Henry's law constant in units of mol L bar, and Pgas is the pressure in bar. -1 Note: Since temperature also affects the solubility of a gas in a liquid, the Henry's law constant is specific to a particular gas at a particular temperature. b. X•10n 1.1546 atm Submit Previous Answers X Incorrect; Try Again; One attempt remaining Part C At 1.01 bar, how many moles of CO2 are released by raising the temperature of 1 litre of water from 20°C to 25° C? Express your answer to four decimal places and include the appropriate units. • View Available Hint(s) μΑ b X•10" 3 7.2 10 mol Submit Previous Answers 國 The solubility of a gas in a liquid increases with increasing pressure. To understand the above statement, consider a familiar example: cola. In cola and other soft drinks, carbon dioxide gas remains dissolved in solution as long as the can or bottle remains pressurized. As soon as the lid is opened and pressure is released, the CO2 gas is much less soluble and escapes into the air. Part B What pressure is required to achieve a CO2 concentration of 0.0480 mol L-1 at 20° C? Express your answer to three significant figures and include the appropriate units. • View Available Hint(s) The relationship between pressure and the solubility of a gas is expressed by Henry's law: Sgas = kH Pgas, Sgas Where is concentration in mol L, kH is the Henry's law constant in units of mol L bar, and Pgas is the pressure in bar. -1 Note: Since temperature also affects the solubility of a gas in a liquid, the Henry's law constant is specific to a particular gas at a particular temperature. b. X•10n 1.1546 atm Submit Previous Answers X Incorrect; Try Again; One attempt remaining Part C At 1.01 bar, how many moles of CO2 are released by raising the temperature of 1 litre of water from 20°C to 25° C? Express your answer to four decimal places and include the appropriate units. • View Available Hint(s) μΑ b X•10" 3 7.2 10 mol Submit Previous Answers 國
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The solubility of a solution was examined for six different temperature settings, shown in the accompanying table. y, Solubility by Weight x, Temperature ( C) 43, 45,...
-
What fraction of monatomic molecules of a gas in a thermal equilibrium possesses kinetic energies differing from the mean value by = 1.0 % and less?
-
At 27C, 10.0 moles of a gas in a 1.50-L container exert a pressure of 130 atm. Is this an ideal gas?
-
Good Morning Food, Inc. is using the profitability index (PI) when evaluating projects. You have to find the PI for the company's project, assuming the company's cost of capital is 14.29 percent. The...
-
A fraction nonconforming control chart is to be established with a center line of 0.01 and two-sigma control limits. (a) How large should the sample size be if the lower control limit is to be...
-
In Exercises determine whether the function is one-to-one. If it is, find its inverse function. f(x) = x 21, x 2
-
\(450 \div 3^{2}-56 \div 2^{2}\) Perform the indicated calculation.
-
Thakin Industries Inc. manufactures dorm furniture in separate processes. In each process, materials are entered at the beginning, and conversion costs are incurred uniformly. Production and cost...
-
The following is information for Monty Corp. for the year ended December 31, 2023: Sales revenue $1,130,000 Loss on inventory due to decline in net realizable value $71,000 Unrealized gain on FV-OCI...
-
GUI Assignment You will be creating a seating arrangement application using JavaFX. The project will comprise of two sections: the interface and the Java code. The application will feature a 3x3...
-
Operationalization is the process of defining the measurement of a phenomenon. It is easier to measure some variables in a straight forward fashion ( i.e. gender). Others are harder to measure. For...
-
Tell us about your culture by analyzing the role that communication plays in your culture. Define and explain your personal, social, and cultural identity. Define intercultural communication...
-
Review this quote before you move on to the identification of your issues: It may very well be that our fundamental ideas about identity and privacy, the strategies that we have collectively pursued,...
-
This week we discussed objectives and goals. We discussed, briefly, the differences between what happening at Twitter and at Mega (Facebook) in terms of layoffs and leadership. In three hundresd...
-
1. When and why has the concept of sustainable development evolved? 2. What is the definition of sustainable development? 3. What are the key features of sustainable development? 4. What elements...
-
Read the 2 articles (Kent State University and Forbes Magazine) below (links provided) and determine one important learning from each article....
-
ealiza una Infografa sobre la diversidad y la Educacin Inclusiva, integrando los siguientes temas: Protocolos interculturales. Principios de la Educacin Inclusiva. Aspectos relacionados con el...
-
Refer to the table to answer the following questions. Year Nominal GDP (in billions) Total Federal Spending (in billions) Real GDP (in billions) Real Federal Spending (in billions) 2000 9,817 578...
-
Ethanol can be used as an automotive fuel either by itself or added to gasoline. Give the equation for the combustion of ethanol.
-
Comet tails always a. Are longest near the sun b. Are shortest near the sun c. Are of constant length d. Point away from a comets direction of motion
-
How does the number of neutrons in a stable nucleus compare with the number of protons? Why is this?
-
Comparative balance sheets and the intervening statement of income for Sears Canada Inc. appear below. Sears Canada is a subsidiary of Sears Holdings Corporation, a U.S. corporation. All amounts are...
-
On January 1, 2016, the U.K. subsidiary of U.S. International Corporation had the following condensed balance sheet, in pounds sterling (in millions): The exchange rate on January 1, 2016 was...
-
Grupo Comercial Chedraui is a food retailer headquartered in Mexico. While it has several subsidiaries, only one is located outside of Mexico. Bodega Latina Co. is a chain of self-service stores...

Study smarter with the SolutionInn App