Prepare this compound from methane gas(CH4) Me HO Prepare this compound from methane gas(CH4) Me HO...
Fantastic news! We've Found the answer you've been seeking!
Question:
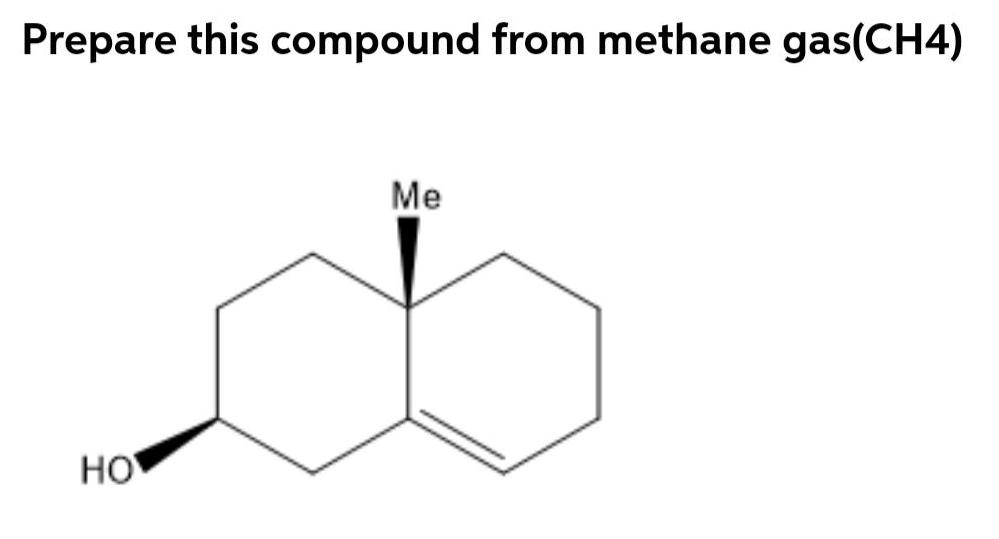
Transcribed Image Text:
Prepare this compound from methane gas(CH4) Me HO Prepare this compound from methane gas(CH4) Me HO
Expert Answer:
Posted Date:
Students also viewed these chemistry questions
-
Methane gas is burned with 30 percent excess air. This fuel enters a steady flow combustor at 101 kPa and 25°C, and is mixed with the air. The products of combustion leave this reactor at 101 kPa...
-
Methane gas is burned completely with 30% excess air at approximately atmospheric pressure. Both the methane and the air enter the furnace at 30(C saturated with water vapor, and the due gases leave...
-
A 50/50 (by mole) gas mixture of methane CH4 and ethylene C2H4 is contained in a cylinder/piston at the initial state 480 kPa, 330 K, 1.05 m3. The piston is now moved, compressing the mixture in a...
-
Write a Digraph client TransitiveClosure whose constructor takes a Digraph as an argument and whose method isReachable (v, w) returns true if there exists some directed path from \(v\) to \(w\), and...
-
How do you define a completion, and how does this definition help a researcher deal with incomplete questionnaires?
-
Innovative Incorporated has recently designed and developed its own payroll system for use in its own business. If there is a market the company may also decide to sell it to outside companies. Work...
-
Your colleague is excited about your good fortune (Problem 3.1) at work, but she only got the promise of a watch or \($300\) cash. You convince her that she will be better in the long run by just...
-
Franciscos mass-produces folding chairs in Mexico. All direct materials are added at the beginning of production, and conversion costs are incurred evenly throughout production. The following...
-
A simple loan with a present value of $5500 will be paid off in 5 years. If the interest rate is 7% what will the future payment be?
-
Why have many institutions moved away from the dormitory concept to smaller rooms occupied by two or three juveniles? allows for separation between types of people who are incarcerated makes the...
-
To make some extra money on the side Ethan decides to join NASA, and is sitting on top of a Falcon X rocket that is waiting to launch into space. When ignited, the rocket takes 7.5 seconds to reach a...
-
Explain why an American call option on a non-dividend-paying stock always has the same price as its European counterpart.
-
A model is reported to be = 5 + 7.2A + 5.8B 2.4C. The standard deviation of each is SY = 2.7, SA = 3.8, SB = 0.58, and SC = 6.5. Which variable, A, B, or C, is most influential in determining Y?
-
What is meant by a standardized regression coefficient? How is it used in regression analysis?
-
NoGrowth Corporation currently pays a dividend of $2.36 per year, and it will continue to pay this dividend forever. What is the price per share if its equity cost of capital is 13% per year?
-
Suppose two stocks have a correlation of 1. If the first stock has an above average return this year, what is the probability that the second stock will have an above average return?
-
If $87,000 is invested in an annuity that earns 5.7%, compounded quarterly, what payments will it provide at the end of each quarter for the next 9 1/2 years? (Round your answer to the nearest cent.)
-
A guitar player can change the frequency of a string by bending itpushing it along a fret that is perpendicular to its length. This stretches the string, increasing its tension and its frequency. The...
-
The future of work may look very different from how it does today. The economy has had a significant influence on OB in recent years. The gig economy (also known as freelancing) has arisen as a major...
-
Creativity is affectively charged, and complex cognitive processes are shaped by both emotions and moods. Creativity may be particularly susceptible to affective influence, mainly because positive...

Study smarter with the SolutionInn App


