What is the goal of a regression model/equation? From the beginning, describe the steps you would...
Fantastic news! We've Found the answer you've been seeking!
Question:
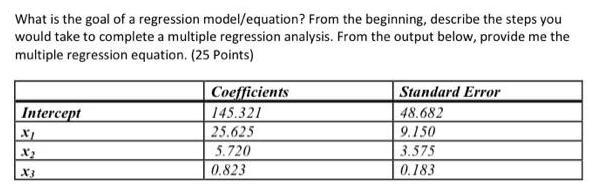
Transcribed Image Text:
What is the goal of a regression model/equation? From the beginning, describe the steps you would take to complete a multiple regression analysis. From the output below, provide me the multiple regression equation. (25 Points) Intercept X₁ X3 Coefficients 145.321 25.625 5.720 0.823 Standard Error 48.682 9.150 3.575 0.183 What is the goal of a regression model/equation? From the beginning, describe the steps you would take to complete a multiple regression analysis. From the output below, provide me the multiple regression equation. (25 Points) Intercept X₁ X3 Coefficients 145.321 25.625 5.720 0.823 Standard Error 48.682 9.150 3.575 0.183
Expert Answer:
Answer rating: 100% (QA)
Based on the output provided the multiple regression equation is Y 145321 25625 X 5720 X2 0823 X3 Th... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The Arnold Company has the following information relating to its inventory for 2009 Beginning inventory 80000 Ending inventory 60000 REQUIRED Create the entries under each of the two systems that...
-
The Webster Store shows the following information relating to one of its products. Inventory, January 1 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .250 units @ $17.00...
-
Nike, Inc., reports the following information relating to its manufacturing activities in the footnotes to its 2018 10-K report: We are supplied by 124 footwear factories located in 13 countries. The...
-
Suppose that a 7% semi-annual coupon bond with a time to maturity of 8 years and a par value of $100 has a price of $106,4. This bond is first callable in 6 years at a redemption price of $104,8....
-
What went wrong with this bankruptcy case?
-
Refer to Example 13.9 and the data in CRIME4.RAW. (i) Suppose that, after differencing to remove the unobserved effect, you think log(polpc) is simultaneously determined with log(crmrte); in...
-
The bank statement for G Greene for the month of March 19X6 is: You are to: (a) Write the cash book up to date, and (b) Draw up a bank reconciliation statement as on 31 March 19X6. 19X6 Mar 1 Dr Cr...
-
Yoklic Corporation currently manufactures a subassembly for its main product. The costs per unit are as follows: Direct materials .......... $ 4.00 Direct labor ................. 30.00 Variable...
-
Sims Company began operations on January 1. Its cost and salesinformation for this year follow.Direct materials$ 30per unitDirect labor$ 50per unitVariable overhead$ 20per unitFixe 2 answers
-
A committee in charge of promoting a Ladies Professional Golf Association tournament is trying to determine how best to advertise the event during the two weeks prior to the tournament. The committee...
-
Analyze the forces in member 1 and 2 for the truss shown in Figure Q4. Assume Area and Modulus of Elasticity are constant for each member. (20 marks) 2 4 m 5 kN 4 m 3 m Figure Q4 The member stiffness...
-
The industrial company Efficar is considering expanding its current product line by manufacturing a new product for which it would benefit from a growing market. This would require investment in new...
-
How does the cultivation of a growth mindset contribute to the development of resilience, and what role does neuroplasticity play in this context ?
-
How does the concept of "post-traumatic growth" illustrate the transformative potential of adversity, and what conditions favor such growth ?
-
Strawberry Fields purchased a tractor at a cost of $37,000 and sold it two years later for $24,500. Strawberry Fields recorded depreciation using the straight-line method, a five-year service life,...
-
What is the coverage of Civil Law and how is it distinct from other sub-categories of law (e.g. Political Law, Commercial Law, and Criminal Law)?
-
can you explain this to me and show me how to work this out. i am struggling with trying to understand how to solve this.
-
5. Convert the following ERD to a relational model. SEATING RTABLE Seating ID Nbr of Guests Start TimeDate End TimeDate RTable Nbr RTable Nbr of Seats RTable Rating Uses EMPLOYEE Employee ID Emp...
-
The atomic masses of 63Li and 73Li are 6.0151 amu and 7.0160 amu, respectively. Calculate the natural abundances of these two isotopes. The average atomic mass of Li is 6.941 amu.
-
(a) The free Cu(I) ion is unstable in solution and has a tendency to disproportionate: Use the information in Table 19.1 to calculate the equilibrium constant for the reaction. (b) Based on your...
-
Determine the standard enthalpy of formation of ethanol (C2H5OH) from its standard enthalpy of combustion (-1367.4 kJ/mol)?
-
Where is the home office of the Association of Certified Fraud Examiners? Would all of its members have an accounting and auditing background? Does the group have a Code of Ethics?
-
Should a forensic accountant express an opinion in a report regarding the guilt or innocence of any person or party?
-
What certificate does the FASNA group issue?

Study smarter with the SolutionInn App


