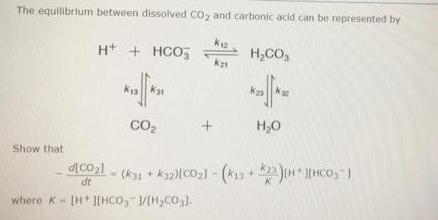
The equilibrium between dissolved CO, and carbonic acid can be represented by H+ + HCO, H,CO,...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The equilibrium between dissolved CO, and carbonic acid can be represented by H+ + HCO, H,CO, CO2 H,0 Show that dt where K- [HJEHCO, /[H,CO,1. Step 1 of 4 The equilibrium between dissolved CO2 and carbonic acid can be represented by K12 H* + HCO5 H2CO3 k21 k31 CO2 H2O Give the expression for CO21 corresponding to the given equilibrium. (Assume that H,0 is incorporated in dt the appropriate equilibrium constants. Use the following as necessary: (CO2), (H*), [HCO,"), [H2CO3). k12. k13, k21, k23, kz1, and k32.) d[CO2) dt Submit The equilibrium between dissolved CO, and carbonic acid can be represented by H+ + HCO, H,CO, CO2 H,0 Show that dt where K- [HJEHCO, /[H,CO,1. Step 1 of 4 The equilibrium between dissolved CO2 and carbonic acid can be represented by K12 H* + HCO5 H2CO3 k21 k31 CO2 H2O Give the expression for CO21 corresponding to the given equilibrium. (Assume that H,0 is incorporated in dt the appropriate equilibrium constants. Use the following as necessary: (CO2), (H*), [HCO,"), [H2CO3). k12. k13, k21, k23, kz1, and k32.) d[CO2) dt Submit
Expert Answer:
Posted Date:
Students also viewed these chemistry questions
-
Show that any positive number M can be represented by (1 + x) / (1 - x), where x lies within the interval of convergence of the series of Problem 11. Hence conclude that the natural logarithm of any...
-
The polarization of a carbonyl group can be represented by a pair of resonance structures: Cyclopropenone and cycloheptatrienone are more stable than anticipated. Cyclopentadienone, however, is...
-
Assume that the core repulsion can be represented by a potential energy of the form Urep = C/rn. So the total potential energy is Use the fact that dU/dr = 0 at r = r0, and the results for Urep at r...
-
State Newtons second law of motion. What are the limitations on the use of Newtons second law? Explain.
-
What is the difference between an unconditional promise to give and an intention to give?
-
The following selected transactions were completed by Bodyworks Co. during October of the current year: Oct. 1. Purchased merchandise from Mantooth Co., $11,800, terms FOB shipping point, 2/10,...
-
Habitat for Humanity International is an ecumenical, faith-based nonprofit dedicated to eliminating substandard housing and homelessness. Habitat constructs, renovates, and repairs houses with the...
-
On January 1, 2014, Wolf Creek Country Club purchased a new riding mower for $15,000. The mower is expected to have a 10-year life with a $1,000 salvage value . What journal entry would Wolf Creek...
-
Nestl S.A. manufactures chocolates in batches. During April 2024, the company produces 5 batches of chocolates, incurring the following costs per batch: Cost Category Batch 1 ($) Batch 2 ($) Batch 3...
-
On a particular day, a random sample of 12 tins of peas was taken from the output of a canning factory, and their contents were weighed. The mean and standard deviation of weights (of the contents of...
-
Bubba just received a gift of $145,756.66 from his childhood friend Bobby. He made a one-time investment 48 years ago at 3.75 percent interest. How much did Bobby originally invest?
-
1.Who determines prices in the free market? What is this economic Concept typically referred to? (the economic concept is of supply and demand ,not the invisible hand ) 2.List & describe the four...
-
In general, do you find that the higher-beta stocks tend to do better in up markets and worse in down markets? Explain.
-
Bodacious Corporation produced 100 units of Product AA. The total standard and actual costs for materials and direct labor for the 100 units of Product AA are as follows:...
-
y The paraboloid is formed by revolving the shaded area around the x axis. Suppose that d = 128 mm and r = 80 mm. The density of the material is p = 5 Mg/m. y = 50x Part A Determine the radius of...
-
Volatility is one of the most prominent terms you'll hear on any trading floor. In financial markets, volatility captures the amount of fluctuation in prices. High volatility is associated to periods...
-
Snells is a retail department store. The following cost-volume relationships were used in developing a flexible budget for the company for the current year. Cost of merchandise sold Selling and...
-
Prairie Outfitters, Inc., a retailer, accepts paymnent through credit cards. During August, credit card sales amounted to $12,000. The processor charges a 3% fee. Assuming that the credit card...
-
Comparative Advantage in the Twenty-First Century. With reference to the theory of comparative advantage, explain how MNEs outsource some services to emerging market economies.
-
Outsourcing and MNEs. How can global outsourcing enable a firm to become a multinational enterprise?
-
Ganados Globalization. After reading the chapters description of Ganados globalization process, how would you explain the distinctions between international, multinational, and global companies?

Study smarter with the SolutionInn App


