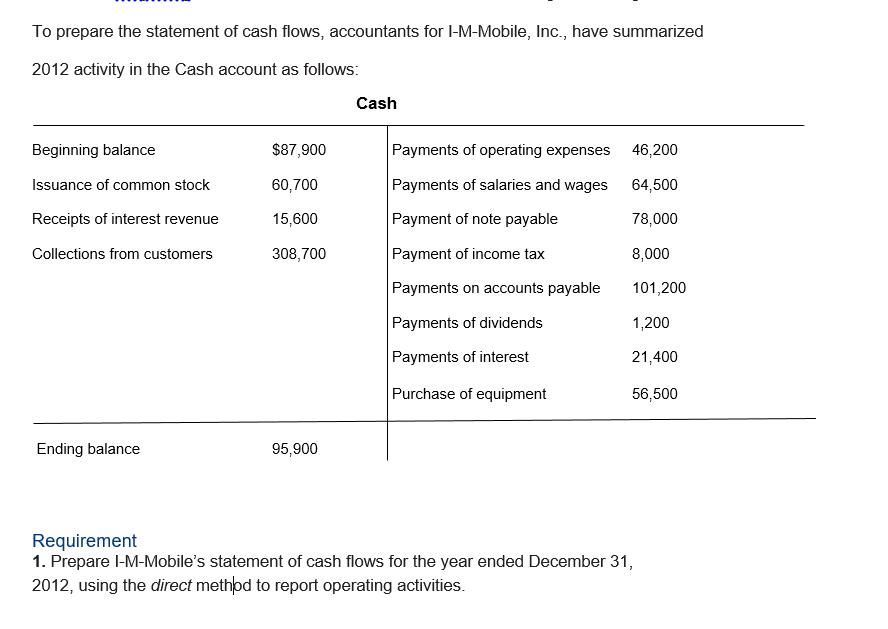
To prepare the statement of cash flows, accountants for I-M-Mobile, Inc., have summarized 2012 activity in...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
To prepare the statement of cash flows, accountants for I-M-Mobile, Inc., have summarized 2012 activity in the Cash account as follows: Beginning balance Issuance of common stock Receipts of interest revenue Collections from customers Ending balance $87,900 60,700 15,600 308,700 95,900 Cash Payments of operating expenses 46,200 Payments of salaries and wages 64,500 Payment of note payable 78,000 Payment of income tax 8,000 Payments on accounts payable 101,200 Payments of dividends 1,200 Payments of interest 21,400 Purchase of equipment 56,500 Requirement 1. Prepare I-M-Mobile's statement of cash flows for the year ended December 31, 2012, using the direct method to report operating activities. To prepare the statement of cash flows, accountants for I-M-Mobile, Inc., have summarized 2012 activity in the Cash account as follows: Beginning balance Issuance of common stock Receipts of interest revenue Collections from customers Ending balance $87,900 60,700 15,600 308,700 95,900 Cash Payments of operating expenses 46,200 Payments of salaries and wages 64,500 Payment of note payable 78,000 Payment of income tax 8,000 Payments on accounts payable 101,200 Payments of dividends 1,200 Payments of interest 21,400 Purchase of equipment 56,500 Requirement 1. Prepare I-M-Mobile's statement of cash flows for the year ended December 31, 2012, using the direct method to report operating activities.
Expert Answer:
Answer rating: 100% (QA)
STATEMENT OF CASH FLOWS DIRECT METHOD Cash flows from operating activities C... View the full answer

Related Book For 

Financial and Managerial Accounting
ISBN: 978-0132497978
3rd Edition
Authors: Horngren, Harrison, Oliver
Posted Date:
Students also viewed these accounting questions
-
To prepare the statement of cash flows, accountants for Four Star Electric Company have summarized 2014 activity in two accounts: Four Star Electrics 2014 income statement and balance sheet data...
-
To prepare the statement of cash flows, accountants for Percy Electric Company have summarized 2016 activity in two accounts: Percy Electric's 2016 income statement and balance sheet data follow:...
-
To prepare the statement of cash flows, accountants for Dora Electric Company have summarized 2012 activity in two accounts as follows: Doras 2012 income statement and balance sheet data follow:...
-
The Louisiana Grill The Louisiana Grill (TLG) is a restaurant in Toronto. TLG is a regional restaurant created and operated by Alex Ven- tresca, a former football player from New Orleans. The company...
-
We offer a guitar at every price point for every skill level, explains Kevin Lello, vice president of marketing at Washburn Guitars. Washburn is one of the most prestigious guitar manufacturers in...
-
The figure shows a plot of vx(t) for a car traveling in a straight line. (a) What is a av, x between t = 6 s and t = 11 s? (b) What is vav, x for the same time interval? (c) What is vav, x for the...
-
An F-test with 5 degrees of freedom in the numerator and 7 degrees of freedom in the denominator produced a test statistic whose value was 5.31. The null and alternate hypotheses were H0: 1 = 2...
-
The Metropolitan Arts Council (MAC) wants to advertise its upcoming season of plays, concerts, and ballets. A television commercial that costs $25,000 will supposedly reach 53,000 potential arts...
-
The accounting records of Crane Corp., a real estate developer, indicated income before income tax of $852,000 for its year ended December 31, 2023, and of $532,000 for the year ended December 31,...
-
The balances in the general ledger accounts of Thunder Graphics are shown below. Instructions On the forms provided in your working papers: 1 . Open the accounts in the general ledger with their...
-
the price elasticity of demand for a product is -.5. When its price is increased by 2%. Quantity demanded will be?
-
The Sarbanes-Oxley Act prohibits public accounting firms from providing cer- tain services to audit clients that are public companies. Which of the following services is not prohibited? a. Internal...
-
What are the general characteristics of diode clamper circuits?
-
Which of the following is not a major threat to an auditor's independence? a. Audit partner's compensation based on obtaining and retaining clients b. Becoming too friendly with the client's...
-
Which of the following statements best explains why the CPA profession has found it essential to promulgate ethical standards and to establish means for en- suring their observance? a. Vigorous...
-
Which of the following would not be a source of information about risk of a po- tential new audit client? a. The previous auditor b. Management c. The Internet d. The new auditor's permanent file
-
D Question 6. Data for two machines P and Q are as shown below. At what MARR will both machines be equally attractive for installation? Initial cost Life in years Inflation per year (for all costs)...
-
Trade credit from suppliers is a very costly source of funds when discounts are lost. Explain why many firms rely on this source of funds to finance their temporary working capital.
-
The following are some of the components included in the master budget. a. Budgeted balance sheet b. Sales budget c. Capital expenditures budget d. Budgeted income statement e. Cash budget f....
-
Thelmas Amusements completed the following transactions during November 2012: Nov 1 Purchased supplies for cash, $700. 4 Purchased inventory on credit terms of 3/10, n/eom, $9,600. 8 Returned half...
-
The adjusted trial balance of Party Piano Tuning Service, Inc., at fiscal year end May 31, 2012, follows. Requirements 1. Prepare Party's 2012 income statement. 2. Prepare the statement of retained...
-
What is reaction coordinate? What is its significance in chemical reaction?
-
Show that for steady state flow process, \(\Delta H=Q-W_{S}\).
-
An ideal gas \(\left(C_{P}=5, C_{V}=3 ight)\) is changed from \(1 \mathrm{~atm}\) and \(22.4 \mathrm{~m}^{3}\) to \(10 \mathrm{~atm}\) and 2.24 \(\mathrm{m}^{3}\) by the reversible process of heating...

Study smarter with the SolutionInn App


