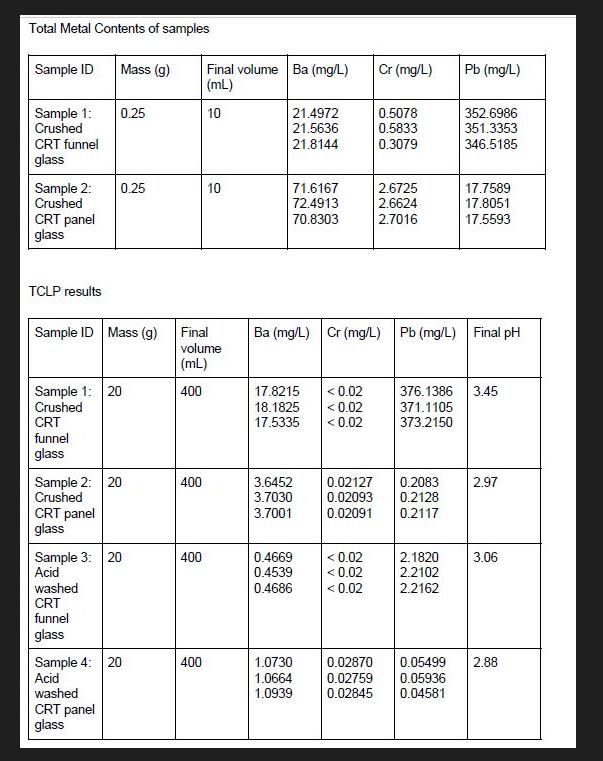
Total Metal Contents of samples Sample ID Sample 1: Crushed CRT funnel glass Sample 2: Crushed...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
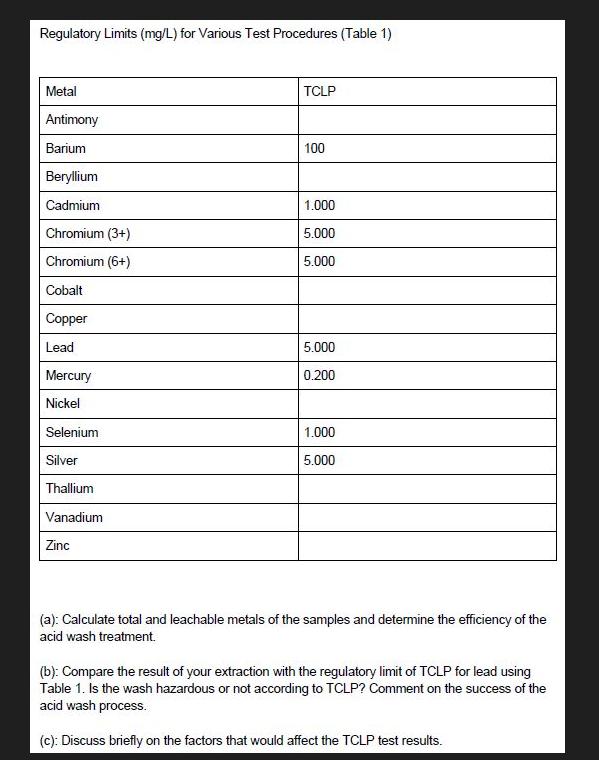
Total Metal Contents of samples Sample ID Sample 1: Crushed CRT funnel glass Sample 2: Crushed CRT panel glass TCLP results CRT funnel glass Sample ID Mass (g) Mass (g) Sample 1: 20 Crushed CRT panel glass 0.25 0.25 Sample 2: 20 Crushed washed CRT funnel glass Sample 3: 20 Acid washed CRT panel glass Sample 4: 20 Acid 400 Final volume (mL) 400 400 Final volume Ba (mg/L) (mL) 10 400 10 21.4972 21.5636 21.8144 71.6167 72.4913 70.8303 17.8215 <0.02 18.1825 <0.02 17.5335 <0.02 3.6452 3.7030 3.7001 0.4669 0.4539 0.4686 1.0730 1.0664 1.0939 Cr (mg/L) Ba (mg/L) Cr (mg/L) Pb (mg/L) Final pH < 0.02 <0.02 <0.02 0.5078 0.5833 0.3079 2.6725 2.6624 2.7016 0.02870 0.02759 0.02127 0.2083 0.02093 0.2128 0.02091 0.2117 Pb (mg/L) 352.6986 351.3353 346.5185 2.1820 2.2102 2.2162 17.7589 17.8051 17.5593 376.1386 3.45 371.1105 373.2150 2.97 3.06 0.05499 2.88 0.05936 0.02845 0.04581 Regulatory Limits (mg/L) for Various Test Procedures (Table 1) Metal Antimony Barium Beryllium Cadmium Chromium (3+) Chromium (6+) Cobalt Copper Lead Mercury Nickel Selenium Silver Thallium Vanadium Zinc TCLP 100 1.000 5.000 5.000 5.000 0.200 1.000 5.000 (a): Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment. (b): Compare the result of your extraction with the regulatory limit of TCLP for lead using Table 1. Is the wash hazardous or not according to TCLP? Comment on the success of the acid wash process. (c): Discuss briefly on the factors that would affect the TCLP test results. Total Metal Contents of samples Sample ID Sample 1: Crushed CRT funnel glass Sample 2: Crushed CRT panel glass TCLP results CRT funnel glass Sample ID Mass (g) Mass (g) Sample 1: 20 Crushed CRT panel glass 0.25 0.25 Sample 2: 20 Crushed washed CRT funnel glass Sample 3: 20 Acid washed CRT panel glass Sample 4: 20 Acid 400 Final volume (mL) 400 400 Final volume Ba (mg/L) (mL) 10 400 10 21.4972 21.5636 21.8144 71.6167 72.4913 70.8303 17.8215 <0.02 18.1825 <0.02 17.5335 <0.02 3.6452 3.7030 3.7001 0.4669 0.4539 0.4686 1.0730 1.0664 1.0939 Cr (mg/L) Ba (mg/L) Cr (mg/L) Pb (mg/L) Final pH < 0.02 <0.02 <0.02 0.5078 0.5833 0.3079 2.6725 2.6624 2.7016 0.02870 0.02759 0.02127 0.2083 0.02093 0.2128 0.02091 0.2117 Pb (mg/L) 352.6986 351.3353 346.5185 2.1820 2.2102 2.2162 17.7589 17.8051 17.5593 376.1386 3.45 371.1105 373.2150 2.97 3.06 0.05499 2.88 0.05936 0.02845 0.04581 Regulatory Limits (mg/L) for Various Test Procedures (Table 1) Metal Antimony Barium Beryllium Cadmium Chromium (3+) Chromium (6+) Cobalt Copper Lead Mercury Nickel Selenium Silver Thallium Vanadium Zinc TCLP 100 1.000 5.000 5.000 5.000 0.200 1.000 5.000 (a): Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment. (b): Compare the result of your extraction with the regulatory limit of TCLP for lead using Table 1. Is the wash hazardous or not according to TCLP? Comment on the success of the acid wash process. (c): Discuss briefly on the factors that would affect the TCLP test results.
Expert Answer:
Answer rating: 100% (QA)
Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment Sample 1 Crushed CRT funnel glass Total metal content Ba 3526986 mgL Cr 05833 mgL Pb 03079 m... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
(a) Calculate total and leachable metals of the samples and determine the efficiency of the acid wash treatment. (b): Compare the result of your extraction with the regulatory limit of TCLP for lead...
-
Question : 1. In the case of digital product development, how to build an ideal and successful Digital Product. Explain and give an example 2. How to define the essentials of User Interaction Design...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1 and 2. On September 1, Irene opened a retail store that specializes in sports car...
-
Walmart Stores has run into opposition when it has tried to open stores in certain towns in the United States. Walmarts capital budgeting process has determined that these locations would be...
-
1. Most aspects of foreign culture, like language, religion, gender roles, and problem-solving strategies, are hard for the casual observer to understand. In what ways do Hollywood movies affect...
-
The passage of the Sarbanes-Oxley Act in the United States has greatly increased the compliance obligations of publicly traded companies. Go online to research how this landmark legislation affected...
-
Compute the (1) current ratio and the (2) quick ratio for Nike, Inc. using the following excerpt from the balance sheet reported in a recent \(10-\mathrm{K}\) of Nike, Inc. At May 31 (in millions)...
-
At December 31, 2011, certain accounts included in the property, plant, and equipment section of Reagan Companys balance sheet had the following balances. Land$230,000 Buildings 890,000 Leasehold...
-
Assume a corporation has just paid a dividend of $ 4.75 pershare. The dividend is expected to grow at a rate of 3.6% per yearforever, and the discount rate is 8.2%. What should be the amountpaid as 2...
-
1. Debug following Echo server and client program. Assume all packages are imported. No indentation errors. Do not focus on upper or lower cases. Hint: Three bugs in each file java file. Below are...
-
I need help with the question above please.. Indicate whether the fol lowing items would appear on the income statement, balance sheet, owner's equity statement or either of two statements: a....
-
A position is an opinion on an issue that can be supported with evidence and reasoning. For example, a position on coal might be "The United States should decrease its reliance on coal power." 2....
-
Give examples of the strengths and weaknesses inherent in using standardsfor planning, controlling, and decision making.How do standards relate to overall cost management?
-
Syntactic operations: English relative clauses For each of the following sentences: Bracket the relative clause Insert a caret (^) in the gap where the relative pronoun originated. Underscore the...
-
In this case study you will be preparing an audit checklist, performing an audit in class, and submitting an audit report for final grading. Details: Before class: . Review case study materials ....
-
Mike's Battery Company has two service departments, Maintenance and Personnel. Maintenance Department costs of $320,000 are allocated on the basis of budgeted maintenance-hours. Personnel Department...
-
You have an investment that includes annual cashflows that areexpected to grow at an annual rate of 0.6% forever and the firstcashflow of $11.50 is expected next year. What is the value of theinves 1...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Sulfamic acid is a primary standard that can be used to standardize NaOH. Sulfamic acid FM 97.094
-
Cocaine metabolism in rats can be studied by injecting the drug and periodically withdrawing blood to measure levels of metabolites by HPLC-mass spectrometry. For quantitative analysis, isotopically...
-
Explain the difference between systematic and random error.
-
The efficient use of assets is indicated by: a. Turnover ratios. b. Debt-related ratios. c. The ratio of debt to equity. d. The ratio of current assets to current liabilities.
-
Which of the following items is not included in the calculation of income from operations? a. Gains on the sale of short-term investments. b. Interest income. c. Interest expense. d. None of the...
-
Hedging (I) There are several schools of thought regarding hedging and its impact on the value of the firm. One school argues that, if the owners of the firm wish to hedge, they can do so in their...

Study smarter with the SolutionInn App


