3. Assign the H NMR spectrum of acetophenone CDCI3 below: 7.97 7.95 7.58 -7.56 7.54 -7.48...
Fantastic news! We've Found the answer you've been seeking!
Question:
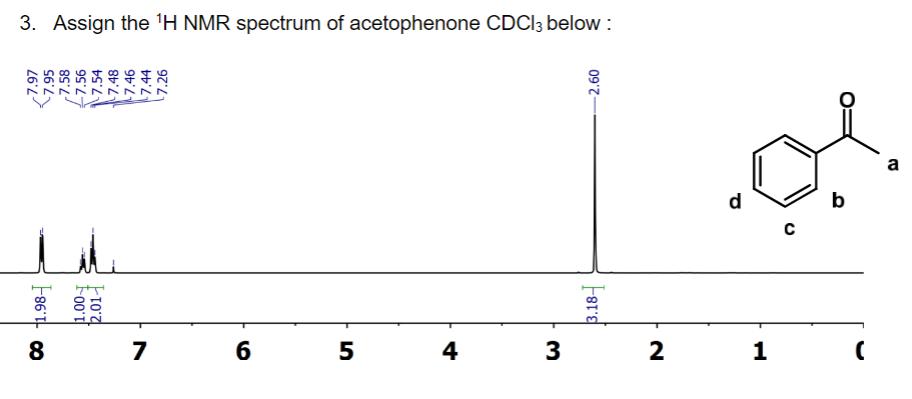
Transcribed Image Text:
3. Assign the ¹H NMR spectrum of acetophenone CDCI3 below: 7.97 7.95 7.58 -7.56 7.54 -7.48 86 CO 8 باشد T'T' ~^7 7 9 5 4 3 2.60 F818 2 d 1 C b B 3. Assign the ¹H NMR spectrum of acetophenone CDCI3 below: 7.97 7.95 7.58 -7.56 7.54 -7.48 86 CO 8 باشد T'T' ~^7 7 9 5 4 3 2.60 F818 2 d 1 C b B
Expert Answer:
Related Book For 

College Accounting
ISBN: 978-1111528126
11th edition
Authors: Tracie Nobles, Cathy Scott, Douglas McQuaig, Patricia Bille
Posted Date:
Students also viewed these chemistry questions
-
The operating activities section of the statement of cash flows for PepsiCo, Inc., owner of the Pepsi beverage brand, is shown as follows: a. Why are impairment and restructuring charges added to the...
-
(7) A compound steel [G = 80 GPa] shaft (Figure P6.16) consists of a solid 55-mm- diameter segment (1) and a solid 40-mm-diameter segment (2). The allowable shear stress of the steel is 70 MPa, and...
-
From station 4+70 with center heights of 1 m in fill, the ground line makes a uniform slope of 4.7% to station 4+135 whose center heights is 2.5m in cut. Determine the slope of the new roadway (%)....
-
Abardeen Corporation borrowed $90,000 from the bank on October 1, 2016. The note had an 8percent annual rate of interest and matured on March 31, 2017. Interest and principal were paid in cash on the...
-
What can a judge do if he or she determines that a piece of legislation is unconstitutional?
-
Four identical particles each with charge Q are at the corners of a square of side L. The particles are released one at a time proceeding clockwise around the square. Each particle is allowed to...
-
Liquid water at \(40^{\circ} \mathrm{F}\) flows down a vertical, thermally insulated, 2 -in. steel pipe. The temperature change of the water is related to its internal energy change by \[...
-
Stevens Co. bought a machine on January 1, 2006 for $875,000. It had a $75,000 estimated residual value and a ten-year life. An expense account was debited on the purchase date. Stevens uses...
-
Conduct investigation into current trends in healthcare using the BU Library search capabilities and any other materials/resources provided to you in the course. Discuss what you think are some of...
-
On January 1, 2010, Phelps Company purchased an 85% interest in Sloane Company for $955,000 when the retained earnings of Sloane Company were $150,000. The difference between implied and book value...
-
A bank offers you 4 different type of interest rate package for your investment. Package I, J 4 =8%; Package II, J 12 =7.5%; Package III, J 2 =8.2%; Package IV, J 365 =7.2%; which of the above...
-
For the following circuit: 2000 5V audio in ' a) If the variable resistor is set to 10k2, what is the current, I, flowing into the base of the transistor when the audio signal is loudest (~1V)? (Note...
-
Blue Sky Corporation, a U.S.-based company, invested 11 million Polish zloty in Green Mountain Company in Lodz, Poland, on January 1, Year 2, when the exchange rate for the Polish zloty (PLN) was...
-
The book value of a firm's equity is $100 million and its market value of equity is $200 million. The face value of its debt is $50 million and its market value of debt is $60 million. What is the...
-
The adjusted trial balance of Eldrich Real Estate Appraisal at June 3 0 , 2 0 1 6 , follows:# ( Click the icon to view the adjusted trial balance. ) Read the requirements. % Requirement 1 . Prepare...
-
Consider the circuit in the diagram where P= 9.10 V and /= 0.596 A. WB A B +1 P 4.00 E C +1 80 WWD 5.00 www 8.00 F At what rate is energy dissipated by the 4.00-Q resistor in the circuit shown? W
-
! Required information PR 2-40 (Algo) Financial Statement Elements; Cost Behavior (LO 2-5, 2-6, 2-8) [The following information applies to the questions displayed below.] Mason Corporation began...
-
1A. If the researcher is concerned about the number of variables, the nature of the analysis, and completion rates, then, he/she is at which stage of the sampling design process (Figure 11.1 in the...
-
Explain the deposit requirement for federal unemployment tax.
-
Following are the columns reflecting the statement of cost of goods manufactured and the income statement from the work sheet of Lambert Motor Corporation as of December 31, the end of the fiscal...
-
Between the end of one month and the 15th day of the next month, the balance in the employers business bank account has been getting smaller and smaller. An employee prepares the next payroll and...
-
Consider the ammonia process in which \(\mathrm{N}_{2}\) and \(\mathrm{H}_{2}\) (with impurities \(\mathrm{Ar}\) and \(\mathrm{CH}_{4}\) ) are converted to \(\mathrm{NH}_{3}\) at high pressure...
-
Consider the simulation flowsheets in Figure 7.36, which were prepared for ASPEN PLUS. The feed stream, S1, is specified, as are the parameters for each process unit. Complete the simulation...
-
Use a process simulator to determine the flow rate of saturated vapor benzene at \(176.2^{\circ} \mathrm{F}\) and \(1 \mathrm{~atm}\) to be mixed with \(100 \mathrm{lbmol} / \mathrm{hr}\) of liquid...

Study smarter with the SolutionInn App



