Under ordinary operation a steady flow of helium gas equal to 50 g/s passes from a large
Question:
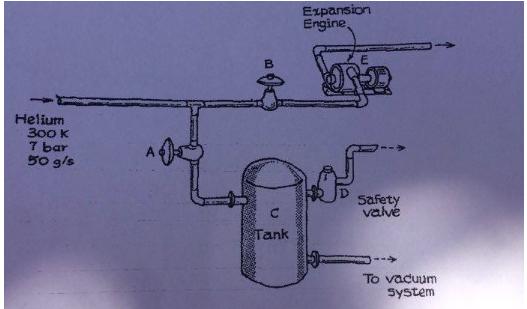
Under ordinary operation a steady flow of helium gas equal to 50 g/s passes from a large storage manifold through an expansion engine as shows in figure. There are, however, certain times when the engine must be shut down for short intervals; the inlet flow cannot, however, be decreased at such times and thus the helium stream must be diverted. It is proposed to emply an adjacent system which at present is not being used. The latter system consists of a large (4m^3) insulated tank (C in figure) with a safety valve venting to the atmosphere through the plant ducting system.
The emergency diverting system is operated in the following manner. If the expansion engine must be shut down (or operates improperly), valve B is shut and A is opened, letting helium into tank C. When the pressure in C rises to 2.8 bar, safety valve D operates, venting gas.
Initially, tank C is evacuated to a very low pressure. We shall assume that when gas enters this tank, it is well-mixed but has negligible heat transfer with the tank walls. Also assume helium is an ideal gas with a constant Cp=20.9 J/molK.
If the safety valve on tank C should operate, we would like to maintain a constant mass of gas in this tank equal to the mass at the time the safety tripped. The massa flow rate into the tank is, as noted above, 50 g/s; the flow rate m* out of the safety valve may be expressed as:
m* = K * A * P/T
where K is a constante, A the valve throat area, and P and T are the pressure and temperature in tank C. To keep the tank mass constante, the valve flow area A will be varied. What are the pressure and temperatura in tank C 10 s after the safety valve opens?
What is the variation of A with time necessary to keep the mass of gas in tank C constant?

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen




