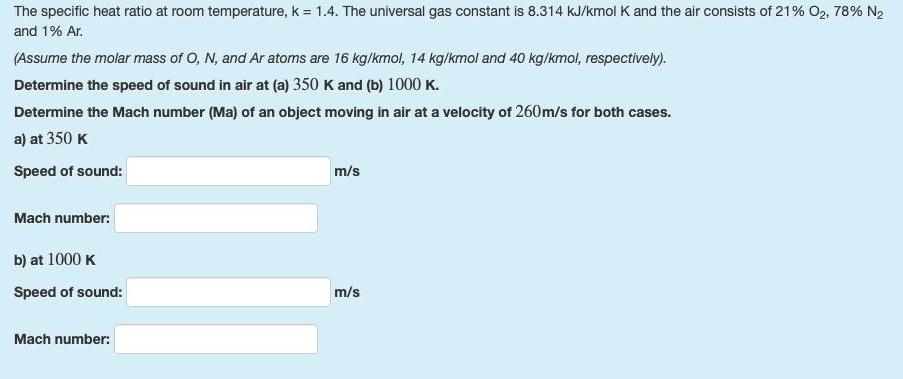
The specific heat ratio at room temperature, k = 1.4. The universal gas constant is 8.314...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The specific heat ratio at room temperature, k = 1.4. The universal gas constant is 8.314 kJ/kmol K and the air consists of 21% O2, 78% N2 and 1% Ar. (Assume the molar mass of O, N, and Ar atoms are 16 kg/kmol, 14 kg/kmol and 40 kg/kmol, respectively). Determine the speed of sound in air at (a) 350 K and (b) 1000 K. Determine the Mach number (Ma) of an object moving in air at a velocity of 260m/s for both cases. a) at 350 K Speed of sound: m/s Mach number: b) at 1000 K Speed of sound: m/s Mach number: The specific heat ratio at room temperature, k = 1.4. The universal gas constant is 8.314 kJ/kmol K and the air consists of 21% O2, 78% N2 and 1% Ar. (Assume the molar mass of O, N, and Ar atoms are 16 kg/kmol, 14 kg/kmol and 40 kg/kmol, respectively). Determine the speed of sound in air at (a) 350 K and (b) 1000 K. Determine the Mach number (Ma) of an object moving in air at a velocity of 260m/s for both cases. a) at 350 K Speed of sound: m/s Mach number: b) at 1000 K Speed of sound: m/s Mach number:
Expert Answer:
Answer rating: 100% (QA)
Calculation of Speed of Sound a at 350 le where r ratio of specific beats V YRT R ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
A steel cylinder of mass 2 kg contains 4 L of liquid water at 25oC at 200 kPa. Find the total mass and volume of the system. List two extensive and three intensive properties of the water
-
A uniform rod AB, of mass 4 kg and length L = 1.5 m, is released from rest in the position shown. Knowing that = 20?, determine the values immediately after release of (a) The angular acceleration...
-
A steel cylinder of mass 2 kg contains 4 L of liquid water at 25 o C at 200 kPa. Find the total mass and volume of the system. List two extensive and three intensive properties of the water.
-
In Problem 12.4 on page 423, you used the percentage of alcohol to predict wine quality. The data are stored in VinhoVerde. From the results of that problem, b 1 = 0.5624 and Sb 1 = 0.1127. a. At the...
-
The Morrison Company produces tennis rackets, the marginal cost of a racket being $20. Because there are many substitutes for the firm's rackets, the price elasticity of demand for its rackets equals...
-
The person who writes his or her name on the back of an instrument. a. bearer ins trument b. blank indors ement c. counterclaim d. dishonorment e. holder in due course f. indorsee g. indorsement h....
-
For a certain incompressible, two-dimensional flow field the velocity component in the \(y\) direction is given by the equation \[ v=3 x y+x^{2} y \] Determine the velocity component in the \(x\)...
-
Buoy manufactures flotation vests in Charleston, South Carolina. Buoys contribution margin income statement for the month ended December 31, 2014, contains the following data: Suppose Overboard...
-
3. The following three-span beam is subjected to two concentrated dead loads as shown below, a uniformly-distributed dead load of 0.8 Kip/ft (including its selfweight), and live load of 1.5 kip/ft...
-
Ryan Ross (111-11-1112), Oscar Omega (222-22-2222), Clark Carey (333-33-3333), and Kim Kardigan (444-44-4444) are equal active members in ROCK the Ages LLC. ROCK serves as agents and managers for...
-
The expected interest rates on one-year bonds for the next 6 years are listed below. i = 6.25%, i=6%, +2=5.5%, i 1+1 = 5.5%, i 1+3 =5.25%, i (+4 1+5 The liquidity premiums for one-to six-year bonds...
-
In 2007, Harvard University rescinded admission to 10 students after reviewing their social media posts.? Do you feel potential employers, current employers, and colleges have the right to access...
-
Consider a VAR of $100 000 for a 95% confidence level. A problem with this information is that while we know that we will lose more than the VAR amount on 5 days out of every 100, we do not know the...
-
Using job order costing, develop an RVU and product cost for the CT procedures shown in the table below. Consider that the CT Department are $10,000,000 ($6,000,000 in direct costs and $4,000,000 in...
-
Timers can generate certain interrupts, so-called overflow and compare match interrupt. Since timer is a peripheral, it can be activated in AVR register. Supposed we need to flash a LED every 100 ms...
-
Why might Nordic bond investors be comfortable with buying unrated bonds issued by a company with no history of sustained profitability?
-
Q-20 Solve the triangle ABC given C = 40, b = 23 cm and a = 19 cm. A-20 Q-21 A-21 97 28 cm 22 cm x
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
Refrigerant-134a at 1 MPa and 100C is throttled to a pressure of 0.8 MPa. Determine the reversible work and exergy destroyed during this throttling process. Assume the surroundings to be at 30C.
-
A refrigeration system operates on the ideal vapor-compression refrigeration cycle with ammonia as the refrigerant. The evaporator and condenser pressures are 200 kPa and 2000 kPa, respectively. The...
-
Refrigerant-134a enters an adiabatic compressor at 2308C as a saturated vapor at a rate of 0.45 m3/min and leaves at 900 kPa and 558C. Determine (a) the power input to the compressor, (b) the...
-
Vertical analysis would rarely be performed on which of the following statements or schedules? a. Income statement b. Adjusting entry worksheet c. Balance sheet d. All of the above are common targets...
-
A statement that lists the assets, liabilities, and stockholders equity of a company in percentages only with no dollar amounts is a a. common-size income statement. b. benchmarking analysis. c....
-
In vertical analysis, the base used for comparison on the income statement is a. total expenses. b. total assets. c. net sales. d. gross profit.

Study smarter with the SolutionInn App


