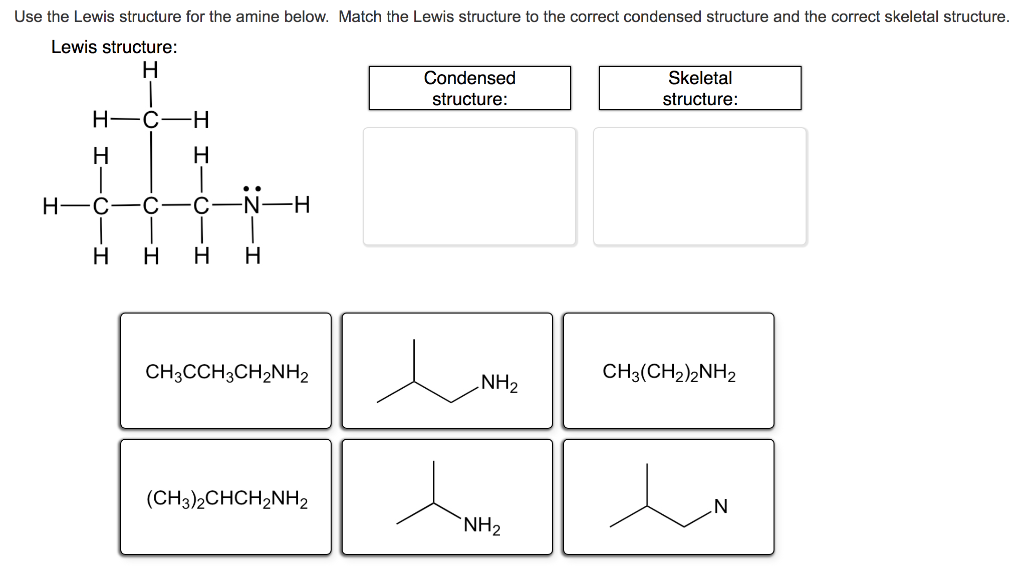
Use the Lewis structure for the amine below. Match the Lewis structure to the correct condensed...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
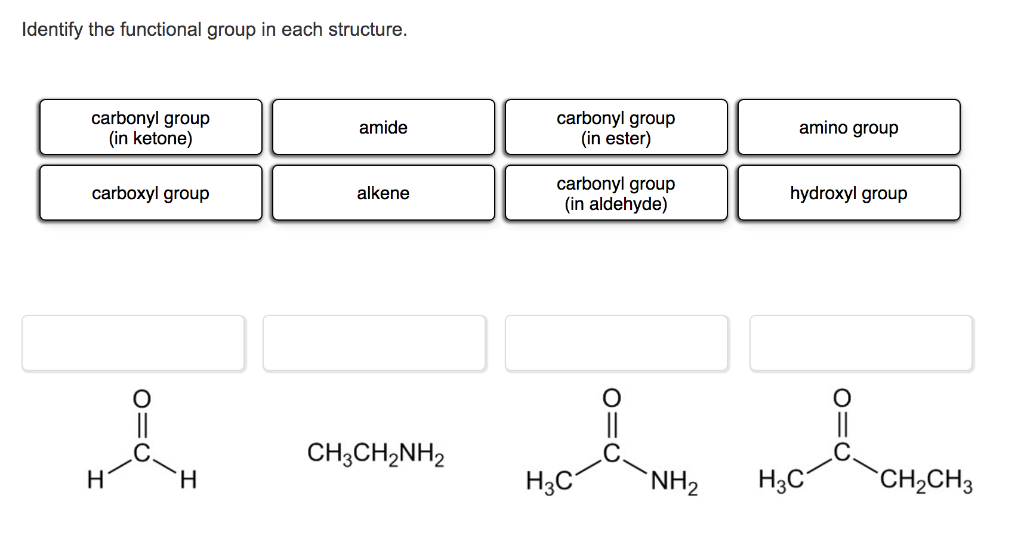
Use the Lewis structure for the amine below. Match the Lewis structure to the correct condensed structure and the correct skeletal structure. Lewis structure: H Condensed Skeletal structure: structure: H-C -H- H H .. H-C C-N-H H H H H CH3CCH3CH2NH2 CH3(CH2)2NH2 NH2 (CH3)2CHCH2NH2 `NH2 N° Identify the functional group in each structure. carbonyl group (in ketone) carbonyl group (in ester) amide amino group carbonyl group (in aldehyde) carboxyl group alkene hydroxyl group CH3CH,NH2 H H. H3C `NH2 H3C `CH2CH3 Which one of the following structures represents an unstable organic compound that is not likely to exist? H H. H H. H. H- C- C-C H H. CH2=CHCH2CH3 CH3CH2CH(CH3)2 Use the Lewis structure for the amine below. Match the Lewis structure to the correct condensed structure and the correct skeletal structure. Lewis structure: H Condensed Skeletal structure: structure: H-C -H- H H .. H-C C-N-H H H H H CH3CCH3CH2NH2 CH3(CH2)2NH2 NH2 (CH3)2CHCH2NH2 `NH2 N° Identify the functional group in each structure. carbonyl group (in ketone) carbonyl group (in ester) amide amino group carbonyl group (in aldehyde) carboxyl group alkene hydroxyl group CH3CH,NH2 H H. H3C `NH2 H3C `CH2CH3 Which one of the following structures represents an unstable organic compound that is not likely to exist? H H. H H. H. H- C- C-C H H. CH2=CHCH2CH3 CH3CH2CH(CH3)2
Expert Answer:
Answer rating: 100% (QA)
Lewis structure H HCH H HCCCNH HHHH carbonyl group in aldehyde 0 O H H H H OH amino group CCCH H ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
The Lewis structure for allene is Make a sketch of the structure of this molecule that is analogous to Figure 9.25. In addition, answer the following three questions: (a) Is the molecule planar? (b)...
-
Consider the Lewis structure for the polyatomic oxyanion shown here, where X is an element from the third period (Na --- Ar). By changing the overall charge, n, from 1 -- to 2 --- to 3 --- we get...
-
Consider the Lewis structure for glycine, the simplest amino acid: (a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each...
-
Parisian Cosmetics Company is planning a one-month campaign for September to promote sales of one of its two cosmetics products. A total of $140,000 has been budgeted for advertising, contests,...
-
Health & Nutrition, Inc., uses the allowance method to account for bad debts. Indicate the effect that each of the following independent transactions will have on gross accounts receivable, the...
-
Give a specic example of data that might be gathered from each of the following business disciplines: accounting, finance, human resources, marketing, information systems, production, and management....
-
A dividend that is paid every quarter or every year is called? a. Regular dividend b. Special dividend c. Property dividend d. Stock dividend
-
Scheduled payments of $1400 due today and $1600 due with interest at 11.5% compounded annually in five years are to be replaced by two equal payments. The first replacement payment is due in 18...
-
12 Match the following concepts with their definitions. Make to Stock Make to Order [Choose ] [Choose ]
-
The CitruSun Corporation ships frozen orange juice concentrate from processing plants in Eustis and Clermont to distributors in Miami, Orlando, and Tallahassee. Each plant can produce 20 tons of...
-
can you explain in simpler terms why variance sum law makes dependent t-tests more powerful
-
Let y = tan(5x + 4). Find the differential dy when x = 1 and dx = 0.4 Find the differential dy when x = 1 and dx: = 0.8
-
Draw the Hasse diagram for the following partial orderings. a. S = {a, b, c} p= {(a, a), (b, b), (c, c), (a, b), (b, c), (a, c)} b. S={a, b, c, d} p = {(a, a), (b, b), (c, c), (d, d), (a, b), (a, c)}...
-
Explain the impact of Sarbanes-Oxley on a company's internal controls. Be sure to define internal controls and discuss the basic principles for assessing internal controls.
-
Calculate the natural frequency of the systems shown in Fig.17.9(a) and (b). W AW W W W W/2 K2 1W/2 W
-
Legislation and regulations are a part of the overall functions of Human Resources. It is essential to learn the differences between these regulations and how that can show up in your career. Analyze...
-
Allows customers to find better alternatives. O a. Red Ocean O b. Cross-selling O c. Exchange O d. Upselling
-
In Problem use absolute value on a graphing calculator to find the area between the curve and the x axis over the given interval. Find answers to two decimal places. y = x 3 ln x; 0.1 x 3.1
-
What is the maximum number of electrons in an atom that can have the following quantum numbers: (a) n = 2, m s = -1/2, (b) n = 5, l = 3, (c) n = 4, l = 3, m 1 = 3; (d) n = 4, l = 0, m 1 = 0?
-
Write the formula for each of the following compounds, being sure to use brackets to indicate the coordination sphere: (a) Hexaamminechromium (III) nitrate (b) Tetraamminecarbonatocobalt (III)...
-
The apparatus shown here has two gas-filled containers and one empty container, all attached to a hollow horizontal tube. When the valves are opened and the gases are allowed to mix at constant...
-
The following information from Jefferson Company's operations is available: Required a. Prepare a multiple-step income statement. Disregard earnings per share amounts. b. Prepare a single-step income...
-
Lincoln Corporation began the year with 50,000 shares of common stock outstanding. On May 1, an additional 18,000 shares of common stock were issued. On July 1, 20,000 shares of common stock were...
-
The following summarized data are related to Kennedy Corporation's operations: Required Prepare a multiple-step income statement for Kennedy Corporation. Include earnings per share disclosure at the...

Study smarter with the SolutionInn App


