Use the Nernst equation to calculate [Ag+] when [Cu2+] = 0.012 M and AEcell 0.410 V....
Fantastic news! We've Found the answer you've been seeking!
Question:
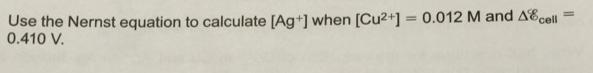
Transcribed Image Text:
Use the Nernst equation to calculate [Ag+] when [Cu2+] = 0.012 M and AEcell 0.410 V. %3D Use the Nernst equation to calculate [Ag+] when [Cu2+] = 0.012 M and AEcell 0.410 V. %3D
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
(a) Use the Nernst equation to write the spontaneous chemical reaction that occurs in the cell in Demonstration 13-1. (b) If you use your fingers as a salt bridge in Demonstration 13-1, will your...
-
The Nernst equation can be applied to half reactions. Calculate the reduction potential at 25oC of each of the following half cells. a. Cu/ Cu2+ (0.10 M) (The half reaction is Cu2+ + 2e n Cu.) b. Cu/...
-
The half-cell reduction potential is provided by the Nernst equation (Equation 14-8): (a) On the graph below, plot the reduction potentials for the FADH2/FAD half-cell (E°² = - 0.219 V) when...
-
Integrate using an appropriate formula For each problem, state the formula number, u and du, identify any constants (if appropriate), and show any constant "adjustments" / "Multiply by 1" if needed....
-
Consider the following information on Stocks I and II: The market risk premium is 7 percent, and the risk-free rate is 4 percent. Which stock has the most systematic risk? Which one has the most...
-
Refer to Table 2.1. Partition G 2 for testing whether the incidence of heart attacks is independent of aspirin intake into two components. Interpret. Table 2.1: Cross-Classification of Aspirin Use...
-
Mr. Prestages foot and lower leg were caught in a combine manufactured by defendant SperryNew Holland. He and his wife sued Defendant for damages arising out of the accident. Their first cause of...
-
A new accounting intern at Gibson Corporation lost the only copy of this periods master budget. The CFO wants to evaluate performance for this period but needs the master budget to do so. Actual...
-
Engineers at electronic company use special wire to manufacture fuzzy logic circuit boards. The wire comes in 1,260 foot rolls that cost $2,700 each. Each board requires 4 1/5 feet of wire. How many...
-
The average owning and operating costs for four types of vehicles in the United States in 2016 include small sedans ($6579), medium sedans ($8604), SUVs ($10,255), and minivans ($9262). Use a Pareto...
-
Monica is a senior financial manager of Eclipse Ltd. She is conducting a capital budgeting analysis on a new product. She has already authorised an extensive market research on the marketability of...
-
Find a set where differentiability is guaranteed and explain why.
-
Consider the following Markov chain: 1 1/2 S 1/2 S. 2 C 1/4 1/4 S 3 1/2 1/2 S 4 1/3 1/3 1/3 1/2 1 S 5 So Given that the above process is in state So just before the first trial, determine by...
-
Your client buys ABC stock for $70. One year later ABC has paid $20 in dividends and your client decides to sell when the stock is at $130. Calculate your clients Holding Period Return.
-
Imagine an experiment where you measure the width, length and height of a box, in centimeters. Additionally, you estimate an uncertainty (error) in each measurement. The goal is to calculate the...
-
Most of the claims the insurance (firms/firm's) processed after the partial mine collapse were categorized as (Worker/Workers') Compensation. Question 10 options: firms ... Worker firms ... Workers'...
-
Electrical power is transmitted across long distances through cables strung from one support to another. The wires are usually made of many smaller wires wrapped into a flexible bundle. For example...
-
(a) What do data breach notification laws require? (b) Why has this caused companies to think more about security?
-
The weak base B (Kb = 1.0 10-5) equilibrates between water (phase 1) and benzene (phase 2). (a) Define the distribution coefficient, D, for this system. (b) Explain the difference between D and K,...
-
Although KOH, RbOH, and CsOH have little association between metal and hydroxide in aqueous solution, Li+ and Na+ doform complexes with OH-: Na + + OH- NaoH (aq) K1 = 0.20 Prepare a table like the...
-
Consider the rechargeable battery: Zn(s) | ZnCl2(aq) || Cl-(aq) | Cl2(l) | C(s) (a) Write reduction half-reactions for each electrode. From which electrode will electrons flow from the battery into a...
-
Flaherty Ltd sells two types of shoes, mens shoes and womens shoes. During the financial year ended 30 June 2019, fixed costs were $460 800 and sales were in the ratio of three units (pairs) of mens...
-
Newbery and Newstead is a public accounting firm specialising in auditing local medium-sized businesses. Fees charged for each audit are determined on the basis of identifiable hours worked on an...
-
Hotbox Ltd produces pizza boxes using two processes cutting and packaging. The production budget for the year ending 30 June 2019 estimated raw materials use of $400 000, factory overhead of $270...

Study smarter with the SolutionInn App



