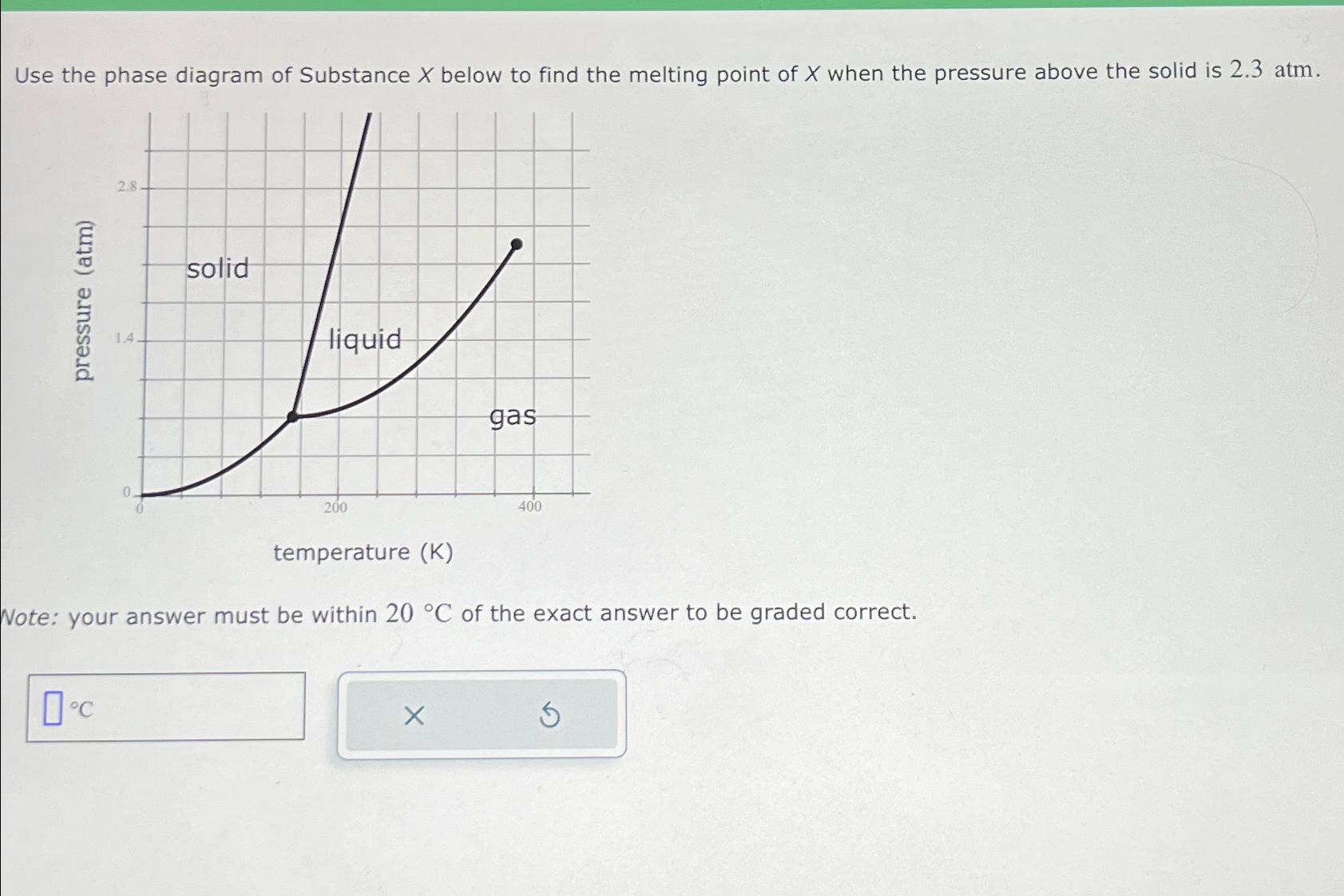
Use the phase diagram of Substance X below to find the melting point of X when...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Use the phase diagram of Substance X below to find the melting point of X when the pressure above the solid is 2.3 atm. pressure (atm) 2.8. 0C 1.4 0 solid liquid 200 gas temperature (K) Note: your answer must be within 20 C of the exact answer to be graded correct. X 400 Use the phase diagram of Substance X below to find the melting point of X when the pressure above the solid is 2.3 atm. pressure (atm) 2.8. 0C 1.4 0 solid liquid 200 gas temperature (K) Note: your answer must be within 20 C of the exact answer to be graded correct. X 400
Expert Answer:
Answer rating: 100% (QA)
To find the melting point of Substance X when the pressure above the solid is 23 atm you need to loo... View the full answer

Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Posted Date:
Students also viewed these chemical engineering questions
-
Specify the characteristics( r , r and ) of a material in which 100MHz uniform plane wave would have a wavelength of 1m, an attenuation of 2Np/m and an intrinsic impedance of 200 ohms.
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Limits from graph In this problem we evaluate limits from the graph. Consider the graph of f given in [Figure 1]. Evaluate each of the following limits (or explain why if the limit does not exist)....
-
Describes the work of leaders and the work of managers. Discuss an example of a new product or service and how it has led from concept to commercial development. Who led the efforts, and how do these...
-
The frozen remains of a young Incan woman were discovered by archeologist Johan Reinhard on Mt. Ampato in Peru during an expedition in 1995. a. How much of the original carbon-14 was present if the...
-
Use the Hubble expansion relation (9.1.1), the temperature scaling relation (9.1.3), and the energy density relation before the electron-positron annihilation (9.3.6b) to show that the temperature as...
-
Puget Sound Divers is a company that provides diving services such as underwater ship repairs to clients in the Puget Sound area. The companys planning budget for May appears below: Required: During...
-
Translate the following business rules into an object relational diagram (ORD) using UMLet (See the above Object Relational Diagram examples). a USER has many ROLES. a USER has one BIRTHDATE. a USER...
-
Selected T-accounts of Moore Company are given below for the just completed year: Required: 1. What was the cost of raw materials put into production during the year? 2. How much of the materials in...
-
Oybean meal is 14% protein; cornmeal is 7% protein. How many pounds of each should be mixed together in order get 280-1b mixture that is 12% protein?
-
5. The density and height of the block shown in Figure Q5 are 750 kg/m and 21 cm respectively. The block floats face down in a fluid whose density is 1660 kg/m. Evaluate the following: (i) The depth...
-
The importance of enforcement of health and safety in the tertiary institution (public and private) and medical laboratories during the times of COVID-19.? include prevention and control of COVID-19...
-
How will you use the knowledge of the SPL model to address contemporary leadership problems in your organization?
-
Start a reflective journal about the Importance of Statement of Work in Project Management. What is Statement of Work (SOW) You must include all points mentioned below. Advantages and core Purpose of...
-
What is printed by the following program? #include void fun (int x) { if (x>0) ( printf("%d", x); fun (x-1); printf("%d", x); } ) int main() { fun (5); return 0; 5 pts
-
You are to examine and discuss about Just In Time( JIT) system that Toyota have adopted to reduce/control their operating costs and to achieve their profitability and non-financial objective. Give an...
-
The overall reaction and equilibrium constant value for a hydrogenoxygen fuel cell at 298 K is 2H 2 (g) + O 2 (g) 2H 2 O(l) K = 1.28 10 83 a. Calculate E cell and G 8 at 298 K for the fuel cell...
-
The chapter opening photograph (page 670) showed how the cobalt(II) chloride equilibrium responded to temperature changes. (a) Look back at that photograph. Is the conversion of the red cation to the...
-
A mixture of butene, C 4 H 8 , and butane, C 4 H 10 , is burned in air to give CO 2 and water. Suppose you burn 2.86 g of the mixture and obtain 8.80 g of CO 2 and 4.14 g of H 2 O. What are the mass...
-
A lithium-ion camera battery is rated at 7500 mAh. That is, it can deliver 7500 milliamps (mA) or 7.5 amps of steady current for an hour. (a) How many moles of electrons can the battery deliver in...
-
Which of the following types of epigenetic changes may promote cancer? a. DNA methylation b. Covalent modification of histones c. Chromatin remodeling d. All of the above may promote cancer.
-
The underlying cause(s) of epigenetic changes associated with cancer may be a. mutations in genes that encode chromatin-modifying proteins. b. environmental agents that alter the function of...
-
Normal (nonmutant) tumor-suppressor genes often function a. as negative regulators of cell division. b. in the maintenance of genome integrity. c. in the stimulation of cell division. d. as both a...

Study smarter with the SolutionInn App


