1- Air undergoes a polytropic compression in a piston-cylinder assembly from p, = 1 bar, T...
Fantastic news! We've Found the answer you've been seeking!
Question:

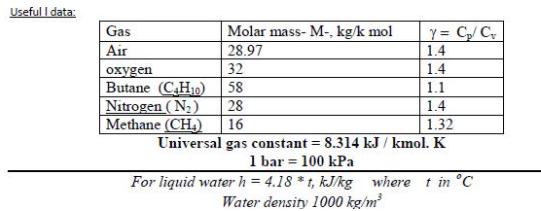
Transcribed Image Text:
1- Air undergoes a polytropic compression in a piston-cylinder assembly from p, = 1 bar, T = 22 °C to p; = 5.5 bars. Employing the ideal gas model, determine the work and heat transfer per unit mass, in kJ/kg, if n= 1.3. Useful I data: Y= C/C 1.4 Gas Molar mass- M-, kg/k mol Air 28.97 32 Butane (CH10) Nitrogen (N) oxygen 1.4 58 1.1 28 1.4 Methane (CH.) 16 1.32 Universal gas constant = 8.314 kJ / kmol. K 1 bar = 100 kPa For liquid waterh = 4.18 *t, kJkg where t in °C Water density 1000 kg/m %3D 1- Air undergoes a polytropic compression in a piston-cylinder assembly from p, = 1 bar, T = 22 °C to p; = 5.5 bars. Employing the ideal gas model, determine the work and heat transfer per unit mass, in kJ/kg, if n= 1.3. Useful I data: Y= C/C 1.4 Gas Molar mass- M-, kg/k mol Air 28.97 32 Butane (CH10) Nitrogen (N) oxygen 1.4 58 1.1 28 1.4 Methane (CH.) 16 1.32 Universal gas constant = 8.314 kJ / kmol. K 1 bar = 100 kPa For liquid waterh = 4.18 *t, kJkg where t in °C Water density 1000 kg/m %3D
Expert Answer:

Related Book For 

Principles of heat transfer
ISBN: 978-0495667704
7th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Posted Date:
Students also viewed these mechanical engineering questions
-
A piston/cylinder contains 2 kg of air at 27C, 200 kPa, shown in Fig. P5.75. The piston is loaded with a linear spring, mass and the atmosphere. Stops are mounted so that V stop...
-
A cylinder containing R-134a at 10C, 150 kPa has an initial volume of 20 L. A piston compresses the R-134a in a reversible, isothermal process until it reaches the saturated vapor state....
-
Determine the rate of heat transfer per unit length from a 5-cm-OD pipe at 150?C placed eccentrically within a larger cylinder of 85% Magnesia wool as shown in the sketch. The outside diameter of the...
-
The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide range of electrophiles. Identify the product that is formed when the conjugate base of diethyl malonate reacts with...
-
Why, in practical terms, is it impossible to separate deductive and inductive approaches to theoretical reasoning?
-
1. Can a company gain advantages besides profit by offshoring? If so, what are they? If not, why not? 2. Does a company face ethical considerations in deciding whether to offshore activities? Why or...
-
Return on Investment, Effect of Depreciation The Chief Operating Officer of the Wisconsin Corporation is considering the effect of depreciation on company ROI. In the most recent year, net operating...
-
The University of Miami bookstore stocks textbooks in preparation for sales each semester. It normally relies on departmental forecasts and preregistration records to determine how many copies of a...
-
profile-image Time remaining: 01 : 59 : 44 Accounting A company began operations on January 1, 2020. Purchases of property, plant and equipment during 2020 was as follows: Cost Residual Value Jan 1,...
-
Part A Walmart Stores (Walmart) is the world's largest retailer. It employs an ''everyday low price'' strategy and operates stores as three business segments: Walmart Stores U.S., International, and...
-
A resistor-inductor-capacitor (RLC) network is depicted in Figure 2. v(t) + L oooo i(t) R vc (t) Figure 2: A resistor-inductor-capacitor (RLC) network. a) Find the differential equation relating the...
-
where q is the reduction in emissions (in pounds of pollutant per day) and C is the daily cost to the firm (in dollars) of this reduction. The government will pay subsidies in the amount of $550 per...
-
The plant manager of Orlando Electronics Company is considering the purchase of new automated assembly equipment. The new equipment will cost $189,000. The manager believes that the new investment...
-
How do historical legacies of colonization, imperialism, and systemic oppression manifest in contemporary structures of power and privilege, and what strategies can be employed to dismantle these...
-
When forecasting pro forma, why did the tutor use (13234/13994)-1 = -5.43 instead of ((13234-13994) / 13994) *100 =-5.45? And why do Net Sales and COGS share the same growth assumption? G5 1 2 fx...
-
3n Compute 4 n=0 iM8 iMa 3n Compute diverg n=0 4
-
{The foiio Irving information appiies to the questions dismayed beio w] The following is the post??closing trial balance for the Whitlow Manufacturing Corporation as of December 31, 2020. Account...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
For the conditions given in Problem 5.4, determine the Nusselt Number and the heat transfer coefficient from Figure.From Problem 5.4: A long cylinder of 0.1 m diameter has a surface temperature of...
-
Compute the average heat transfer coefficient hc for 10?C water flowing at 4 m/s in a long, 2.5-cm-ID pipe (surface temperature 40?C) by three different equations and compare your results. Also...
-
Determine the steady-state temperatures of two radiation shields placed in the evacuated space between two infinite planes at temperatures of 555 K and 278 K. The emissivity of all surfaces is 0.8....
-
A lessor entered into a 5-year lease appropriately classified as a sales-type lease. The cost of the underlying asset was \(\$ 40,000\) and the fair value of the asset was \(\$ 50,000\). The lease...
-
Using the information from Exercise 17-72, complete the following requirements from the lessor's perspective. Required a. Determine the classification of the lease to Try-Star Leasing Company. b....
-
Lessory Corporation, a manufacturer of equipment, enters into a lease of specialized equipment with LesseeX Corp. Title to the asset remains with Lessory Corp upon lease expiration. LesseeX Corp does...

Study smarter with the SolutionInn App


