3- Five kg of butane (C,H) in a piston-cylinder assembly undergo a process from p, =...
Fantastic news! We've Found the answer you've been seeking!
Question:

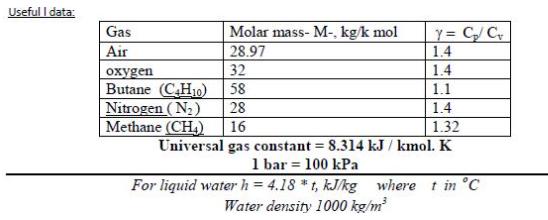
Transcribed Image Text:
3- Five kg of butane (C,H) in a piston-cylinder assembly undergo a process from p, = 5 MPa, T1 = 500 K to p2 = 3 MPa, T =450 K during which the relationship between pressure and specific volume is pv" = constant. Determine the work, in kJ. Useful I data: Y = C/C 1.4 1.4 Gas Molar mass- M-, kg/k mol 28.97 Air 32 oxygen Butane (CH10) Nitrogen (N2) Methane (CH) 58 1.1 28 1.4 16 1.32 Universal gas constant = 8.314 kJ / kmol. K 1 bar = 100 kPa For liquid water h = 4.18 * t, kJ/kg where t in °C Water density 1000 kg/m 3- Five kg of butane (C,H) in a piston-cylinder assembly undergo a process from p, = 5 MPa, T1 = 500 K to p2 = 3 MPa, T =450 K during which the relationship between pressure and specific volume is pv" = constant. Determine the work, in kJ. Useful I data: Y = C/C 1.4 1.4 Gas Molar mass- M-, kg/k mol 28.97 Air 32 oxygen Butane (CH10) Nitrogen (N2) Methane (CH) 58 1.1 28 1.4 16 1.32 Universal gas constant = 8.314 kJ / kmol. K 1 bar = 100 kPa For liquid water h = 4.18 * t, kJ/kg where t in °C Water density 1000 kg/m
Expert Answer:

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these mechanical engineering questions
-
Nitrogen at 200oC, 300 kPa is in a piston cylinder, volume 5 L, with the piston locked with a pin. The forces on the piston require a pressure inside of 200 kPa to balance it without the pin. The pin...
-
Air contained within a piston cylinder assembly undergoes three processes in series: Process 1-2: Compression at constant pressure from P1=10 lbf/in2, V1= 4ft3 to state 2. Process 2-3:...
-
The relationship between pressure and temperature in saturated steam can be expressed as:* where Y = pressure and t = temperature. Using the method of nonlinear least squares (NLLS), obtain the...
-
If you were able to dictate economic policy, how would you strengthen the automatic stabilizers in this country? Why would your solutions work?
-
How much would have to be paid on an invoice for $328 with terms of 2/10 ROG if the merchandise invoice is dated January 3, the merchandise arrives January 8, and the invoice is paid (a) January 11;...
-
How would you attempt to (a) Estimate the crystal field stabilization energy of FeF 2 , (b) Determine the overall stability constant of [Co(NH 3 ) 6 ] 3+ in aqueous solution given that the overall...
-
AutoAnswer, Inc., a manufacturer of telephone answering machines, uses a factory overhead applied rate to charge overhead costs to its manufactured products. The company manager estimates that...
-
Consider trade relations between the United States and Mexico. Assume that the leaders of the two countries believe the payoffs to alternative trade policies are as follows: a. What is the dominant...
-
Using the provided financial statements for Church & Dwight Co., calculate their Fiscal 2021 Net Sales forecast if we assume Net Sales will increase at the same percentage growth rate as Fiscal 2019...
-
The bond equivalent yield of a 91-day T-bill is 5%. What is the price of the bill for a $10,000 face value?
-
Each year a school runs a health check to monitor the heights of the students. For each student the height is recorded in centimeters. The average height is 150 centimeters, with an SD of 10...
-
Four particles with charges of 7.7 C, 3.4 C, 1.8 C, and -7.5 C are placed at the corners of a 2.4 m x 2.4m square. Determine the potential difference between the center of the square and infinity....
-
From the following information prepare a monthly cash budget for the three months ending 31st Dec.2020 Sales Materials Wages Production Admin. Selling, etc Month (RM) (RM) (RM) (RM) (RM) June 6,000...
-
Brittany is changing the tire of her car on a steep hill 20.0 m high. She trips and drops the 10.0-kg spare tire, which rolls down the hill with an initial speed of 2.00 m/s and then rolls us the...
-
Normandeau Company's actual indirect cost pool amounted to $1,400,000 and the direct labour cost pool was $5,400,000. Overhead is allocated on the basis of direct labour hours. Actual and budgeted...
-
Find the derivative of f(x)=sin(5x) f'(x) =
-
Home Products, Inc. (HPI) is a leading manufacturer of prescription and ethical drugs; specialty foods and candies; and proprietary drugs. Important product names include Advil, Anacin, Dimetapp,...
-
Solve each problem. Find the coordinates of the points of intersection of the line y = 2 and the circle with center at (4, 5) and radius 4.
-
Water from the combustion of hydrogen and pure oxygen is at 3800 K and 50 kPa. Assume we only have H2O, O2, OH and H2 as gases with the two simple water dissociation reactions active find the...
-
Methanol, CH3OH, is burned with 200% theoretical air in an engine and the products are brought to 100 kPa, 30C. How much water is condensed per kilogram of fuel?
-
Two hydraulic piston/cylinders are connected with a line. The master cylinder has an area of 5 cm2 creating a pressure of 1000 kPa. The slave cylinder has an area of 3 cm2. If 25 J is the work input...
-
Microsoft, Inc., recognizes revenue from its licensed software products at the time of sale or time of installation. Despite receiving such revenue in cash, Microsoft defers the recognition of a...
-
In September 2015, Boeing agreed to terms to sell 300 planes to China in one of the largest long-term airline contracts ever signed. With contracts to sell a plane, a company such as Boeing would...
-
On April 14, 2006, The Wall Street Journal carried an article headlined Research in Motion Skeptics Pipe Up. The article concerned Research in Motion Ltd. (RIM), the manufacturer of the BlackBerry....

Study smarter with the SolutionInn App


