Using the figure on the following page on how to achieve an octet for cations and...
Fantastic news! We've Found the answer you've been seeking!
Question:

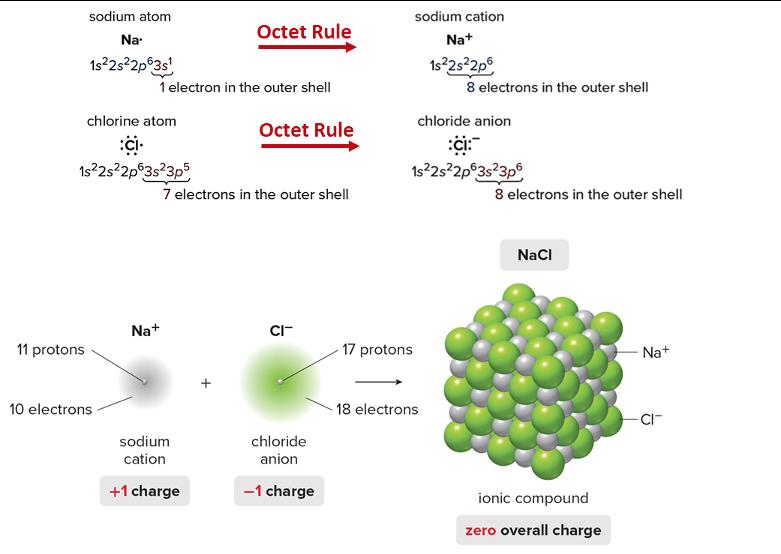
Transcribed Image Text:
Using the figure on the following page on how to achieve an octet for cations and anions, please draw a Lewis dot structure for each of the following elements. Also, identify what net charge each element would adopt to achieve the valence electron configuration of a noble gas (achieve a stable octet). ***See Lithium below as an example. Li Lit where it has 1+ net charge a. Example: Li Lewis Structure: b. F Lewis structure: c. Mg Lewis structure: d. Cl Lewis Structure: e. K Lewis structure: f. S Lewis structure: g. Ca sodium atom Na- 1s²2s²2p63s¹ 11 protons chlorine atom :CI 1s²2s²2p63s²3p5 10 electrons 1 electron in the outer shell Na+ Octet Rule sodium cation +1 charge Octet Rule 7 electrons in the outer shell CI- chloride anion -1 charge sodium cation Na+ 1s²2s²2p6 17 protons chloride anion :ci: 1s²2s²2p63s²3p6 8 electrons in the outer shell 18 electrons 8 electrons in the outer shell NaCl ionic compound zero overall charge Na+ -CI- Using the figure on the following page on how to achieve an octet for cations and anions, please draw a Lewis dot structure for each of the following elements. Also, identify what net charge each element would adopt to achieve the valence electron configuration of a noble gas (achieve a stable octet). ***See Lithium below as an example. Li Lit where it has 1+ net charge a. Example: Li Lewis Structure: b. F Lewis structure: c. Mg Lewis structure: d. Cl Lewis Structure: e. K Lewis structure: f. S Lewis structure: g. Ca sodium atom Na- 1s²2s²2p63s¹ 11 protons chlorine atom :CI 1s²2s²2p63s²3p5 10 electrons 1 electron in the outer shell Na+ Octet Rule sodium cation +1 charge Octet Rule 7 electrons in the outer shell CI- chloride anion -1 charge sodium cation Na+ 1s²2s²2p6 17 protons chloride anion :ci: 1s²2s²2p63s²3p6 8 electrons in the outer shell 18 electrons 8 electrons in the outer shell NaCl ionic compound zero overall charge Na+ -CI-
Expert Answer:
Answer rating: 100% (QA)
Solution z atomic number a Li15251 Lewis 23 5 b F H0 23 pps Lewis 29 ... View the full answer

Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Posted Date:
Students also viewed these chemistry questions
-
Draw a Lewis dot structure for each of the following compounds: a. CH 3 CH 2 OH b. CH 3 CN
-
Draw a Lewis dot structure for each of the following atoms: (a) Carbon (b) Oxygen (c) Fluorine (d) Hydrogen (e) Bromine (f) Sulfur (g) Chlorine (h) Iodine
-
The figure on the following page shows a unit circle, which will be used to illustrate a number of the subtasks. (A) For the angle a, sin(a)= -13/20 and - < < -1/2-. Mark a in a hot circle. (Use a...
-
Suppose that \(\mathrm{x}\) is a linked-list Node. What is the effect of the following code fragment? \(t\). next \(=x\). next; \(x \cdot\) next \(=t ;\)
-
In addition to the facts covered in Questions 3 through 5, you learn that the banini is not freely convertible into dollars. The Varoom foreign-exchange and tax laws provide that a foreign-invested...
-
The Big Club has total assets of $150,000, current assets of $80,000 (current assets includes $30,000 of cash, $10,000 of short-term investments, $20,000 of accounts receivable, and $20,000 of...
-
P 55 Use the Standard Normal Table or technology to find the z-score that corresponds to the cumulative area or percentile. Table 4-Standard Normal Distribution Arca Z 0 Z .09 .08 .07 .06 .05 .04 .03...
-
Elizabeth (Liz) Lake lives in a million-dollar mansion in Don Mills. She spends time with her three children at an 1134-square-metre waterfront cottage in central Ontario. The attractive widow also...
-
A 38 kg crate rests on a floor. A horizontal pulling force of 170 N is needed to start the crate moving. 1.What is the coefficient of static friction between the crate and the floor? 2.The same crate...
-
Consolidation related simulation example: Millennium Capital Management, Inc., (MCM) acquired a 90% interest in NextGen, Inc. MCM's Financial Manager, Matthew Steven, has prepared a draft memo to the...
-
Determine the size of the required equal payments that are to be made in 6 months, 12 months and 1 2 years from today to pay off a loan of $169,902.00 borrowed today at 6% p.a. interest? (Focal point...
-
Write the equation of the line with a slope of -2 and a y-intercept of 3 .
-
5 Given $ 100 deg/s = Q = 45 deg y = 360 deg * = 10 deg/s + = 10 deg/s 0 = 5 deg Find the body axis roll, pitch, and yaw rates using the kinematic equations.
-
The XYZ Company issued discount debt bonds with the nominal value of $100,000.The bonds have a maturity period of 5 year and a coupon rate of 6% annually.Calculate the issue price of the bonds taking...
-
How did the United States shift from a nation of agrarian farmers to an industrial powerhouse during the late 19th and early 20th centuries?
-
Wayne Company is located at 99 Fifth Avenue New York City, NY, 10001. The company is a general partnership using the calendar year and accrual basis for both book and tax purposes. It engages in the...
-
You are the financial manager for a large organization, and two managers have presented the following proposals. Each proposal requires a $100,000 investment, and the cost of capital (discount rate)...
-
Compile data on consumption and expenditures for the following categories in 30 different countries: (1) food and beverages, (2) clothing and footwear, (3) housing and home operations, (4) household...
-
Resolution with HPLC Teaching Assistant, an Excel spreadsheet that simulates high performance liquid chromatography separations. Download the Excel file from SaplingPlus, macmillanlearning.com, or...
-
What is the purpose of the WO3 WO 3 and Cu Cu in Figure 27-8? Figure 27-8 Leee Oxygen WO oxidation catalyst Cu reduction catalyst and O scavenger 7. Encapsulated sample Ceramic crucible Helium...
-
Select the fluorescent derivatization reagent from 1 to 3 for each of the following applications from the literature. a. Quantitation of thiols such as glutathione, cysteine, homocysteine, and y y...
-
How did the Fed hit its target for the federal funds rate in the pre-2008 scarce-reserves regime?
-
Draw a demand and supply graph for the federal funds market to show the demand and supply of reserves in the current ample-reserves regime. How can the Fed increase the federal funds rate? Show the...
-
What is the zero lower bound in monetary policy? What tools can the Fed use if it wants to implement an expansionary monetary policy at the zero lower bound?

Study smarter with the SolutionInn App


