The correct sequence of increasing covalent character is represented by (A) LiCl < NaCI < BeCl,...
Fantastic news! We've Found the answer you've been seeking!
Question:
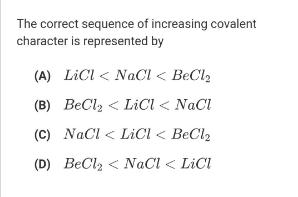
Transcribed Image Text:
The correct sequence of increasing covalent character is represented by (A) LiCl < NaCI < BeCl, (B) BeClz < LiCl < NaCl (C) NaCl < LiCl < BeCl2 (D) BeClz < NaCl < LiCI The correct sequence of increasing covalent character is represented by (A) LiCl < NaCI < BeCl, (B) BeClz < LiCl < NaCl (C) NaCl < LiCl < BeCl2 (D) BeClz < NaCl < LiCI
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A system is represented by a block diagram as shown in Figure E3.5. Write the state equations in the form of Equations (3.16) and (3.17). FIGURE E3.5 Block diagram.
-
The remainder operator _____________. a. Is represented by a forward slash b. Must follow a division operation c. Provides the quotient of integer division d. Is none of the above
-
The wing of a small plane is represented by a simplified prismatic cantilever beam model acted on by the distributed loads shown in the figure. Assume constant EI = 1200 kN*m 2 . Find the tip...
-
Randi Corp. is considering the replacement of some machinery that has zero book value and a current market value of $3,700. One possible alternative is to invest in new machinery that costs $30,900....
-
A copper bar AB of length 25 in. and diameter 2 in. is placed in position at room temperature with a gap of 0.008 in. between end A and a rigid restraint (see figure). The bar is supported at end B...
-
1. There are three sections of English 101. In Section I, there are 25 students, of whom 5 are mathematics majors. In Section II, there are 20 students, of whom 6 are mathematics majors. In Section...
-
Use the all-possible-regressions selection on the fuel consumption data in Table B.18. Perform a thorough analysis of the best candidate models. Compare your results with stepwise regression....
-
Duval Ford, LLC, sold a new Ford F-250 pick-up truck to David Sweat. Before taking delivery, Sweat ordered a lift kit to be installed on the truck by a Duval subcontractor. Sweat also replaced the...
-
1. Explain the transfer of energy from the water through the dam to become electricity that can be used in your home. ( In your response, be sure to identify sources of both potential and kinetic...
-
You talk to Sally about getting paid for the work you're doing. You suggest $25 an hour and she agrees. You are only doing this temporarily since you have some extra time so you set yourself up as a...
-
To what extent is Fuyao a value creator or a value destroyer, and what are the potential sources of that creation (or destruction)? Is dividend policy irrelevant to Fuyao? If so, why? If not, why?...
-
Assess and then rank how persuasive each of the following subject lines are to try to persuade staff to help out with the company's fundraising efforts. (1 mark each)* 3 points Very Persuasive...
-
In this excerpt from the memoirs of Ann Carson, she described leaving her husband because he did not see her as his equal. She also wrote about her efforts to go into business for herself. As you...
-
1. From your point of view, is there an ethical issued involved in this case? In your own words, explain why there is or is not an ethical issued involved and what the issue is. 2. Is the financial...
-
The following reserve table appeared in the financial statements of Brown Company. (in thousands of barrels) Year ended Dec. 31 2015 2016 2017 170 240 325 Beginning of year Revisions of previous...
-
What are the intricate mechanisms underlying the hypothalamic-pituitary-adrenal axis and its role in stress response regulation ?
-
Sarah wants to buy a $600000 one-bedroom apartment in Toronto. She believes that she can make a down payment of $160000. The terms of the mortgage are a 5.0% per year compounded monthly for a fixed 3...
-
Recall that Chapter 8 described the binary search algorithm for finding a particular entry in an ordered list. The idea behind binary search is to begin looking in the exact center of the list. If...
-
As a group, the noble gases are very stable chemically (only Kr and Xe are known to form compounds). Use the concepts of shielding and the effective nuclear charge to explain why the noble gases tend...
-
Write Lewis structures for the reaction What kind of bond joins Al and Cl in the product? AICH + Cl- AICla
-
A chemist in the nineteenth century prepared an unknown substance. In general, do you think it would be more difficult to prove that it is an element or a compound? Explain.
-
In problem 49, when does the estate's taxable year end? problem 49, John Henry died on May 1, 2018. When does his final 1040 have to be filed?
-
John Henry died on May 1, 2018. When does his final 1040 have to be filed?
-
John and Mary Hughes file their income tax return on a calendar year basis. John dies on May 15, 2018. Mary remarries on July 4, 2018. Can Mary file a joint return with John for the taxable year 2018?

Study smarter with the SolutionInn App



