In the amplifier circuit shown in Figure 3 the input resistance R= 100 0, R. =...
Fantastic news! We've Found the answer you've been seeking!
Question:
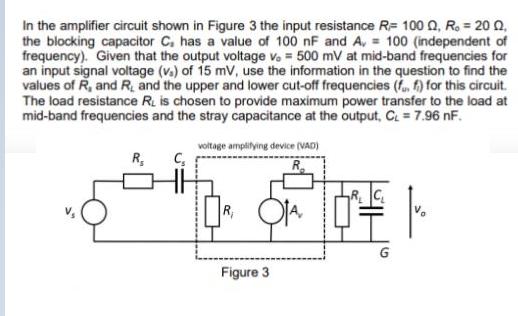
Transcribed Image Text:
In the amplifier circuit shown in Figure 3 the input resistance R= 100 0, R. = 20 Q, the blocking capacitor C. has a value of 100 nF and A, = 100 (independent of frequency). Given that the output voltage v. = 500 mV at mid-band frequencies for an input signal voltage (v.) of 15 mV, use the information in the question to find the values of R, and R, and the upper and lower cut-off frequencies (f, f) for this circuit. The load resistance R. is chosen to provide maximum power transfer to the load at mid-band frequencies and the stray capacitance at the output, C = 7.96 nF. voltage amplilying device (VAD) R, R R, Figure 3 In the amplifier circuit shown in Figure 3 the input resistance R= 100 0, R. = 20 Q, the blocking capacitor C. has a value of 100 nF and A, = 100 (independent of frequency). Given that the output voltage v. = 500 mV at mid-band frequencies for an input signal voltage (v.) of 15 mV, use the information in the question to find the values of R, and R, and the upper and lower cut-off frequencies (f, f) for this circuit. The load resistance R. is chosen to provide maximum power transfer to the load at mid-band frequencies and the stray capacitance at the output, C = 7.96 nF. voltage amplilying device (VAD) R, R R, Figure 3
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these electrical engineering questions
-
A $100,000 GNMA pass-through bond issue has a value of $113,830. The value of the interest-only payments is $46,921. What is the value of the principal-only payment?
-
The following reaction has a value of Go = -2.1Kj/mol (-0.50 kcal/mol). CH3Br + H2S CH2 SH + HBr (a) Calculate Keq at room temperature (25 C) for this reaction as written. (b) Starting with a 1 M...
-
A van der Waals gas has a value of z = 1.00061 at 410. K and 1 bar and the Boyle temperature of the gas is 195 K. Because the density is low, you can calculate V m from the ideal gas law. Use this...
-
Distinguish between a National Health Insurance system and a National Health Service. Provide examples of each. What kind of a system does the United States have?
-
Without generating the Sensitivity Report, adapt the sensitivity analysis procedure presented in Secs. 7.1 and 7.2 to conduct the sensitivity analysis specified in the four parts of Prob. 9.2-22.
-
Natural frequency of aluminum bar given by consistent-mass matrices a. \(58554 \mathrm{rad} / \mathrm{s}\) lumped-mass matrices b. \(33806 \mathrm{rad} / \mathrm{s}\) consistent-mass matrices c....
-
What rules apply to service and filing of pleadings?
-
The following information is available for Pacilio Security Services Inc. for 2011, its first year of operations. Pacilio provides security services for local sporting events. The following summary...
-
Calculating the Days' Payable Period . The following information is taken from the annual report of Coca-Cola Enterprises, Inc.: (amounts in millions) Year 2 Year 1 Net revenue $18,706 $18,158 Cost...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
1. What has included the complete set of financial statements? (Please list them only; the explanation is not required) 2. Fair presentation requires the faithful representation of the effects of...
-
Canada is considered a developed country. Does that mean we have reached the end of the economic development road? yes or no, why ? Or can you think of any specific achievements we could collectively...
-
Observations with what types of missing values should not be ignored because any analysis that includes the variables with these values will be biased?
-
James is currently on a bridge, 2 1 meters above the water and throws a ball in the horizontal direction. The ball gets to the water surface at a horizontal distance of 1 2 meters from James. ( a )...
-
Sam has two jobs, one for the winter and one for the summer. In the winter, he works as a lift attendant at a ski resort where he earns $13 per hour. During the summer, he drives a tour bus around...
-
Vera Spencer, a divorced mother of one, lived in a house she owned in suburban Mississauga, Ontario. Her daughter Karen was sixteen years old at the time of the events in question. Vera had an...
-
Problem 1 (10 pts) In the leg extension exercise, the quadriceps mechanism applies a muscle force M through the patellar tendon at the point P as shown (we are only considering one leg). Solve for...
-
Will the prediction interval always be wider than the estimation interval for the same value of the independent variable? Briefly explain.
-
A pair of standard dice are rolled. What is the probability of observing the following: a. The sum of the dice is equal to 7. b. The sum of the dice is equal to 9. c. The sum of the dice is less than...
-
2.25 moles of an ideal gas with C V ,m = 5/2 R is transformed from an initial state T 680. K and P = 1.15 bar to a final state T = 298.K and P = 4.75 bar. Calculate U, H, and S for this process.
-
The reaction of nitric oxide (NO(g)) with molecular hydrogen (H2(g)) results in the production of molecular nitrogen and water as follows: 2NO(g) + 2H 2 (g) N 2 O(g) + 2H 2 O(g) The experimentally...
-
An auditor accesses a magnetic disk file that contains the dollar amounts of all client inventory items by style number. The information on the disk is in no particular sequence. By use of a...
-
An auditor's client has a magnetic disk that contains the detail of its customers' insurance policies by policy number. Unknown to the auditor is the fact that many of the policies are for...
-
In many cases, the auditor uses the output of a computer-assisted audit program for the same purpose as if the output were obtained through manual means. For each of the following examples of...

Study smarter with the SolutionInn App


