Show your work for your calculations. Mass of crucible + lid Mass crucible + lid+ hydrate...
Fantastic news! We've Found the answer you've been seeking!
Question:
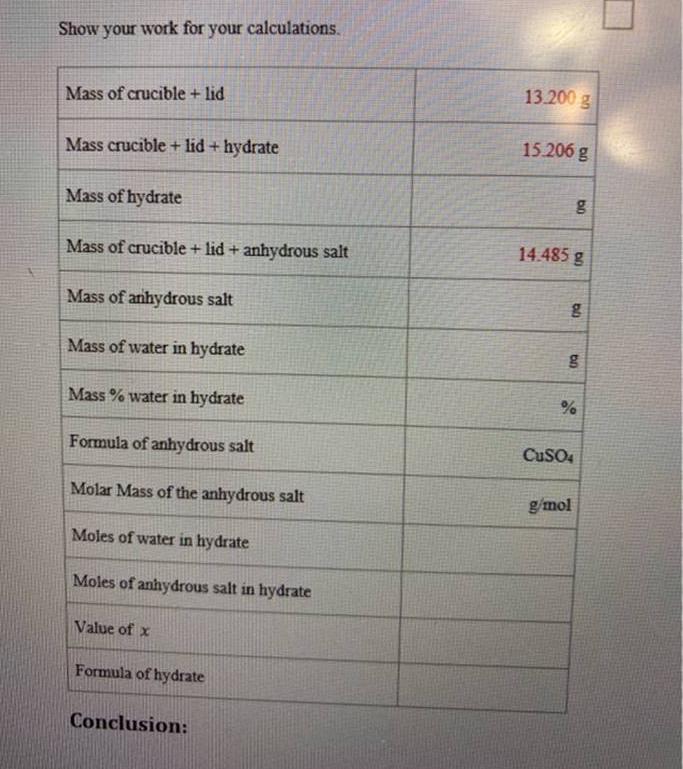

Transcribed Image Text:
Show your work for your calculations. Mass of crucible + lid Mass crucible + lid+ hydrate Mass of hydrate Mass of crucible + lid + anhydrous salt Mass of anhydrous salt Mass of water in hydrate Mass % water in hydrate Formula of anhydrous salt Molar Mass of the anhydrous salt Moles of water in hydrate Moles of anhydrous salt in hydrate Value of x Formula of hydrate Conclusion: 13.200 g 15.206 g bo 14.485 g g g % CuSO4 g/mol Questions: 1. True or False: The hydrate is the salt that remains after heating. 2. True or False: The anhydrous salt does not contain water. 3. The hydrate salt was heated and the weight after heating was determined to be 3.894. Is this the mass of the hydrate salt or of the anhydrous salt? 4. Given the following grams of the substances, what is the percentage of C in the compound? • Mass of C = 2.402 g Mass of the compound = 10.304 g 5. Given the following grams of the elements, what is the empirical formula of the compound? • 2.45 g Si 12.4 g Cl Show your work for your calculations. Mass of crucible + lid Mass crucible + lid+ hydrate Mass of hydrate Mass of crucible + lid + anhydrous salt Mass of anhydrous salt Mass of water in hydrate Mass % water in hydrate Formula of anhydrous salt Molar Mass of the anhydrous salt Moles of water in hydrate Moles of anhydrous salt in hydrate Value of x Formula of hydrate Conclusion: 13.200 g 15.206 g bo 14.485 g g g % CuSO4 g/mol Questions: 1. True or False: The hydrate is the salt that remains after heating. 2. True or False: The anhydrous salt does not contain water. 3. The hydrate salt was heated and the weight after heating was determined to be 3.894. Is this the mass of the hydrate salt or of the anhydrous salt? 4. Given the following grams of the substances, what is the percentage of C in the compound? • Mass of C = 2.402 g Mass of the compound = 10.304 g 5. Given the following grams of the elements, what is the empirical formula of the compound? • 2.45 g Si 12.4 g Cl
Expert Answer:
Answer rating: 100% (QA)
Here are the stepbystep calculations with explanations Mass of crucible lid 13200 g Mass crucible li... View the full answer

Related Book For 

University Physics with Modern Physics
ISBN: 978-0321501219
12th Edition
Authors: Hugh D. Young, Roger A. Freedman, Lewis Ford
Posted Date:
Students also viewed these accounting questions
-
A certain magnesium sulfate salt hydrate sample has a mass of 246.506 mg. After heating and cooling the sample has a new mass of 120.366 mg. How many moles of water were included in 1 mole of the...
-
A solution of 1.00 g of anhydrous aluminum chloride, AlCl3, in 50.0 g of water freezes at 21.11C. Does the molar mass determined from this freezing point agree with that calculated from the formula?...
-
Water of mass m = 20 g is enclosed in a thermally insulated cylinder at the temperature of 0 C under a weightless piston whose area is S = 410 cm2 . The outside pressure is equal to standard...
-
Historically, investment spending has experienced more extreme upward and downward swings than consumer spending. Why do you think this is so?
-
What is an audit adjustment, and why is the resolution of such adjustments important to audit quality? What role should professional skepticism play when management disagrees with the auditor about...
-
Distinguish among warehouses, distribution centers, and cross-docking facilities.
-
Jason Corporation has only common stock outstanding. The firm reported earnings per share of \(\$ 4.00\) for the year. During the year, Jason paid dividends of \(\$ 0.85\) per share. At year end, the...
-
The bookkeeper for Geronimo Company has prepared the following balance sheet as of July 31, 2014. The following additional information is provided. 1. Cash includes $1,200 in a petty cash fund and...
-
6.9 through 6.12 For the beam and loading shown, consider section n-n and determine (a) the largest shearing stress in that section, (b) the shearing stress at point a. 15,15 30 15 15 20 0.5 m 72 kN...
-
The comparative balance sheet of Iglesias Inc. for December 31, 20Y3 and 20Y2, is as follows: Additional data obtained from an examination of the accounts in the ledger for 20Y3 are as follows: a....
-
Make a report based on the questions below: 1. Decide the type of business that is being carried out. (can be in the form of a product or service), preferably the one you understand about the...
-
This self-assessment consists of 12 questions. For each question, type your brief answer in the space provided. After answering all 12 questions, compare what you wrote with the correct answer along...
-
A sedan weighing 2,000 kg is traveling at 120 kph over a series of sinusoidal bumps having a roughness height of 6 cm and spacing of 3 m. The spring at each wheel has a stiffness of 100,000 N/m. The...
-
wlamart argues that it provides valuable jobs to small communities and should be allowed to grow. critics argue that these jobs are often low paying, with few benefits. what is your position? is a...
-
Joint ventures have significant advantages as well several disadvantages versus a wholly owned subsidiary with regard to investment abroad. Define the two types of investment opportunities and...
-
Equipment has a debit side in a trial balance, why here they are in credit side?
-
Assume today is 01/01/98. The yield compounding frequency is annual. A risk free pure discount bond with face value $1,000 maturing on 12/31/98 is selling today at $940.The forward rate for the...
-
Select a mass spectrometric technique with the highest mass resolution for identifying an unknown compound being eluted from a liquid chromatography column
-
A sled starts from rest at the top of a hill and slides down with a constant acceleration. At some later time it is 14.4 m from the top; 2.00 s after that it is 25.6 m from the top, 2.00s later 40.0...
-
Certain rifles can fire a bullet with a speed of 965 m/s just as it leaves the muzzle (this speed is called the muzzle velocity). If the muzzle is 70.0 cm long and if the bullet is accelerated...
-
The two vectors A and B are drawn from a common point, and C = 1 + B. (a) Show that if C2 = A2 + B2, the angle between the vectors A and B is 90. (b) Show that if C2 < A2 + B2, the angle between the...
-
Argue that for undirected graphs, the connected components algorithm does find the connected component of the given initial vertex.
-
Prove that for directed graphs, the connected components algorithm finds the set of vertices that can be reached from a given initial vertex \(v\). Prove that this set is a closed set (see Example...
-
For an undirected graph with the adjacency matrix below, find the connected components. \[ \left(\begin{array}{llllllllll} 0 & 1 & 0 & 0 & 0 & 0 & 0 & 0 & 0 & 1 \\ 1 & 0 & 0 & 0 & 0 & 0 & 0 & 0 & 0 &...

Study smarter with the SolutionInn App


