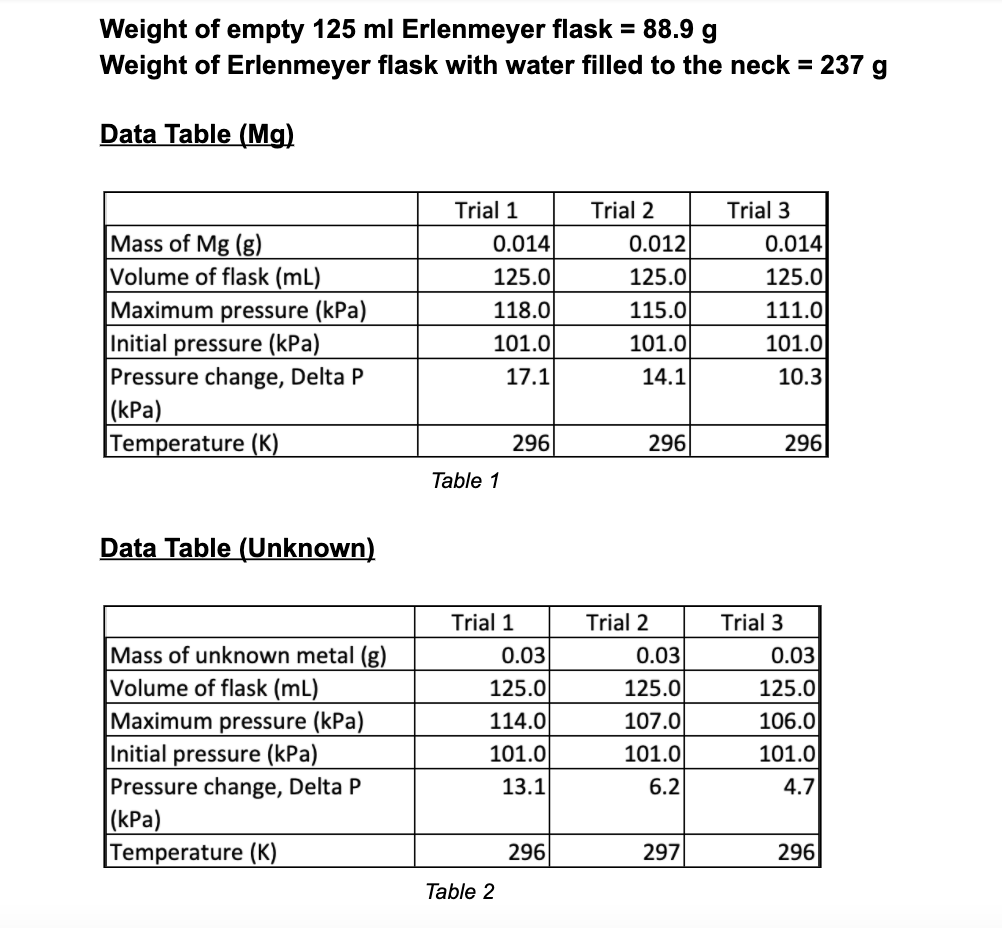
Weight of empty 125 ml Erlenmeyer flask = 88.9 g Weight of Erlenmeyer flask with water...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
Weight of empty 125 ml Erlenmeyer flask = 88.9 g Weight of Erlenmeyer flask with water filled to the neck = 237 g Data Table (Mg) Mass of Mg (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Data Table (Unknown) Mass of unknown metal (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Trial 1 0.014 125.0 118.0 101.0 17.1 Table 1 296 Trial 1 0.03 125.0 114.0 101.0 13.1 Table 2 296 Trial 2 0.012 125.0 115.0 101.0 14.1 Trial 2 296 0.03 125.0 107.0 101.0 6.2 297 Trial 3 0.014 125.0 111.0 101.0 10.3 296 Trial 3 0.03 125.0 106.0 101.0 4.7 296 Data Analysis: 1. Using the measured weight of the empty 125 mL weight when filled with water, and the known density of water at the temperature of the measurement, calculate the "effective" volume of the 125 mL Erlenmeyer flask, i.e., the volume available for occupancy by the H₂ gas. (You must also take into account the volume of the added HCl solution, which decreases the volume available to the H2 gas.) 2. Calculate the molar amounts of each piece of magnesium and unknown metal that you used. ENEW 3. Based on the measured temperatures and pressure changes for each reaction, use the ideal gas the molar amount of hydrogen gas that was produced in each reaction. 4. Calculate the volume occupied by one mole of hydrogen gas (molar volume) at STP. 5. Compare your calculated molar volume, at STP, with the accepted molar volume of an ideal gas at STP, viz., 22.41 L/mol. If the values do not compare well, suggest possible sources of experimental error. 6. Use your calculated value for the STP molar volume to determine the atomic weight of your unknown metal. What is the likely identity of the metal? Weight of empty 125 ml Erlenmeyer flask = 88.9 g Weight of Erlenmeyer flask with water filled to the neck = 237 g Data Table (Mg) Mass of Mg (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Data Table (Unknown) Mass of unknown metal (g) Volume of flask (ml) Maximum pressure (kPa) Initial pressure (kPa) Pressure change, Delta P (kPa) Temperature (K) Trial 1 0.014 125.0 118.0 101.0 17.1 Table 1 296 Trial 1 0.03 125.0 114.0 101.0 13.1 Table 2 296 Trial 2 0.012 125.0 115.0 101.0 14.1 Trial 2 296 0.03 125.0 107.0 101.0 6.2 297 Trial 3 0.014 125.0 111.0 101.0 10.3 296 Trial 3 0.03 125.0 106.0 101.0 4.7 296 Data Analysis: 1. Using the measured weight of the empty 125 mL weight when filled with water, and the known density of water at the temperature of the measurement, calculate the "effective" volume of the 125 mL Erlenmeyer flask, i.e., the volume available for occupancy by the H₂ gas. (You must also take into account the volume of the added HCl solution, which decreases the volume available to the H2 gas.) 2. Calculate the molar amounts of each piece of magnesium and unknown metal that you used. ENEW 3. Based on the measured temperatures and pressure changes for each reaction, use the ideal gas the molar amount of hydrogen gas that was produced in each reaction. 4. Calculate the volume occupied by one mole of hydrogen gas (molar volume) at STP. 5. Compare your calculated molar volume, at STP, with the accepted molar volume of an ideal gas at STP, viz., 22.41 L/mol. If the values do not compare well, suggest possible sources of experimental error. 6. Use your calculated value for the STP molar volume to determine the atomic weight of your unknown metal. What is the likely identity of the metal?
Expert Answer:
Answer rating: 100% (QA)
Experiment 1 Mg Trial 1 Mass of Mg 0014 g Maximum pressur... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Water enters a pump at 100 kPa with a mass flow rate of 20 kg/s and exits at 500 kPa with the same mass flow rate. If the density () of water is 1000 kg/m3, determine (a) the net rate of flow work...
-
The mass of the Empty Flask is 49.6536gand the Density is assumed to be 1. Ill fipet Tlalibiration. True Volume Appeck ent alPpRalent Mass of mos5 of pipet scadig (m1) 0,00 1000 Plask (5) 4A 6536...
-
Measured data for pressure versus volume during the compression of a refrigerant within the cylinder of a refrigeration compressor are given in the table below. Using data from the table complete the...
-
An air-filled X-band rectangular waveguide has dimensions a = 2.286 cm and b = 1.016 cm. If the waveguide has copper walls ( = o , = o , = 5.8 x 10 7 S/m), find the attenuation in dB/m due to the...
-
Corporation X realized a material gain when its facilities at a designated floodway were acquired by the urban renewal agency. How should the gain be reported in the income statement?
-
Why do developed countries such as the United States regulate trade through the mechanisms described in this section? Why might a less developed country do so? To promote fair trade the WTO prohibits...
-
What is meant by the expectation gap?
-
Doughboy Bakery would like to buy a new machine for putting icing and other toppings on pastries. These are now put on by hand. The machine that the bakery is considering costs $90,000 new. It would...
-
Make sure your answer is completely correct please.Thanks YOS Corporation is expected to pay a dividend of \( \$ 2.80 \) next year. If the current stock price is \( \$ 51.00 \) and the cost of equity...
-
Stang Corporation issued to Bradley Company $400,000 par value, 10-year bonds with a coupon rate of 12 percent on January 1, 20X5, at 105. The bonds pay interest semiannually on July 1 and January 1....
-
A cash discount is usually a discount allowed for: Multiple choice question. buying on credit late payment after the scheduled due date prompt payment payment before the end of the discount period
-
1 . A 3 0 - year 6 . 5 0 0 % semi - annual coupon bond has a tenor of 1 7 years and a yield to maturity of 7 . 7 5 0 % . In percent of PAR, what is the price of the bond today? 2 . A 3 0 - year 5 . 5...
-
1. Explain how a country could have an absolute disadvantage in the production of a product relative to another country and still have a comparative advantage in the production of the product. 2....
-
Kubin Company's relevant range of production is 14,000 to 20,500 units. When it produces and sells 17,250 units, its average costs per unit are as follows: Average Cost per Unit Direct materials $...
-
Use the methods of accurately determining dimensions of materials for construction projects by scaling requirements from drawings and specifications and calculate the area and no of lining material...
-
Liberty Ventures Inc. reported $350,400 profit in 2023 and declared preferred dividends of $17,800. The following changes in common shares outstanding occurred during the year: Jan. 1 130,000 common...
-
Explain Stanley Milgram's research on obedience. What did his study's findings reveal about human behavior? Is it possible to repeat this study today? Why or why not? What is the foot-in-the-door...
-
Grace is training to be an airplane pilot and must complete five days of flying training in October with at least one day of rest between trainings. How many ways can Grace schedule her flying...
-
When 0.273 g of Mg is heated strongly in a nitrogen (N2) atmosphere, a chemical reaction occurs. The product of the reaction weighs 0.378 g. Calculate the empirical formula of the compound containing...
-
Greenhouse gases absorb (and trap) outgoing infared radiation (heat) from Earth and contribute to global warming. The molecule of a greenhouse gas either possesses a permanent dipole moment or has a...
-
Match each of the elements on the right with its description on the left: (a) A pale yellow gas Nitrogen (N2) that reacts with water. Boron (B) (b) A soft metal that reacts Aluminum (Al) with water...
-
Describe a mutual fund prospectus.
-
You just inherited $4,000. You need to decide whether to invest this money in a mutual fund, pay down your credit card debt, or simply hold the money in an interest-bearing checking account to pay...
-
Describe the characteristics of a closed-end, open-end, load, and no-load mutual fund.

Study smarter with the SolutionInn App


