We've dealt with the molecular diagram of diatomic molecules. However, conceptually the problem is much more...
Fantastic news! We've Found the answer you've been seeking!
Question:
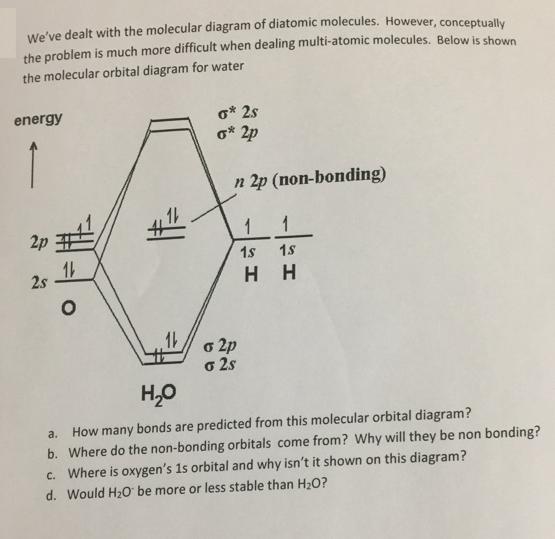
Transcribed Image Text:
We've dealt with the molecular diagram of diatomic molecules. However, conceptually the problem is much more difficult when dealing multi-atomic molecules. Below is shown the molecular orbital diagram for water o* 2s o* 2p energy n 2p (non-bonding) 1 1 2p 1s 18 2s H H o 2p o 2s H,0 How many bonds are predicted from this molecular orbital diagram? b. Where do the non-bonding orbitals come from? Why will they be non bonding? c. Where is oxygen's 1s orbital and why isn't it shown on this diagram? a. d. Would H2o be more or less stable than H2O? We've dealt with the molecular diagram of diatomic molecules. However, conceptually the problem is much more difficult when dealing multi-atomic molecules. Below is shown the molecular orbital diagram for water o* 2s o* 2p energy n 2p (non-bonding) 1 1 2p 1s 18 2s H H o 2p o 2s H,0 How many bonds are predicted from this molecular orbital diagram? b. Where do the non-bonding orbitals come from? Why will they be non bonding? c. Where is oxygen's 1s orbital and why isn't it shown on this diagram? a. d. Would H2o be more or less stable than H2O?
Expert Answer:
Answer rating: 100% (QA)
a four bonds are predicted from this molecular orbital diag... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
How do 2p orbitals differ from each other? How do 2p and 3p orbitals differ from each other? What is a nodal surface in an atomic orbital?
-
When an electron occupies a 2s orbital on an N atom it has a hyperfine interaction of 55.2 mT with the nucleus. The spectrum ofN02 shows an isotropic hyperfine interaction of 5.7 mT. For what...
-
Why do we hybridize atomic orbitals to explain the bonding in covalent compounds? What type of bonds form from hybrid orbitals, or ? Explain.
-
Day and Night formed an accounting partnership in 2014. Capital transactions for Day and Night during 2014 are as follows: Partnership net income for the year ended December 31, 2014; is $68,400...
-
From the partial worksheet for Rose Co. shown in Figure prepare a formal incomestatement. ROSE CO PARTIAL WORKSHEET FOR YEAR ENDED DECEMBER 31, 201X income Statement Account Titles Dr Cr 5000 295000...
-
In Exercises 25, use the relative frequency histogram to (a) Identify the class with the greatest, and the class with the least, relative frequency. (b) Approximate the greatest and least relative...
-
Flowchart symbols represent what aspect of a system?
-
Worf Co. both purchases and constructs various equipment it uses in its operations. The following items for two different types of equipment were recorded in random order during the calendar year...
-
For this assignment, you will be assigned eight (8) specific ingredients from four different products. You will be given four questions in this quiz, and for each question, you will be asked to...
-
Consider the two mutually exclusive projects in Table P5.49. Salvage values represent the net proceeds (after tax) from disposal of the assets if they are sold at the end of each year. Both projects...
-
Project Cost NPV A $1,061,191 $122,737 B 561,758 58,102 C 1,647,849 280,660 D 1,026,020 89,365 E 191,870 17,568 F 1,333,625 76,960 G 3,102,642 123,240 H 275,568 79,367 I 2,044,070 60,506 J 1,017,567...
-
The city of Moscow, Idaho has decided to build a 3-story high school on the edge of town (next to "open terrain"). There is an Intermediate reinforced masonry, 25 foot wide, shear wall parallel to...
-
3. Why is acetone soluble in water and dichloromethane? Ci H C. H CI H dichloromethane acetone 4. Circle the compound that is most soluble in water. A. H B. H H 5. Circle the organic compound that is...
-
Black, an engineer, is injured while visiting the White Manufacturing Company. Black was splattered in the face with hot metal from a die-casting machine which he was observing at the time. What...
-
Last year Minden Company introduced a new product and sold 2 5 , 8 0 0 units of it at a price of $ 9 3 per unit. The product's variable expenses are $ 6 3 per unit and its fixed expenses are $ 8 3 1...
-
The Noohi case on enforceability of an arbitration provision. (1) what went on in the case, i.e., what were the basic facts and what was the major issue that the court decided, and (2) what was the...
-
0.75' C D a 4.5' N 0.5' B 10.5' 2.5' X
-
If a process has a six-sigma capability, what is the process capability index? a. 1 b. 2 c. 6 d. 12
-
In an experiment to study the photoelectric effect, a scientist measures the kinetic energy of ejected electrons as a function of the frequency of radiation hitting a metal surface. She obtains the...
-
Write a balanced equation for the reaction of each of the following compounds with water: (a) SO2(g) (b) Cl2O7(g) (c) Na2O2(s) (d) BaC2(s) (e) RbO2(s) (f) Mg3N2(s) (g) NaH(s).
-
Indicate whether each statement is true or false. If it is false, rewrite it so that it is true. (a) If you measure the rate constant for a reaction at different temperatures, you can calculate the...
-
C. Gomez, corporate engineer of Allied Products Ltd. is proposing the replacement of existing equipment with more efficient equipment. According to information received by Gomez, the proposed...
-
Match each of the following descriptions, a through \(h\), with a capital budgeting method: (1) payback method, (2) net present value, (3) profitability index, or (4) internal rate of return. More...
-
SavingsPlus Inc. is considering an investment in computer technology. The project would require an initial investment of \(\$ 240,000\) and have an expected life of six years with no salvage value....

Study smarter with the SolutionInn App


