What are the masses of the four different BrCl molecules? Express the masses in atomic mass...
Fantastic news! We've Found the answer you've been seeking!
Question:
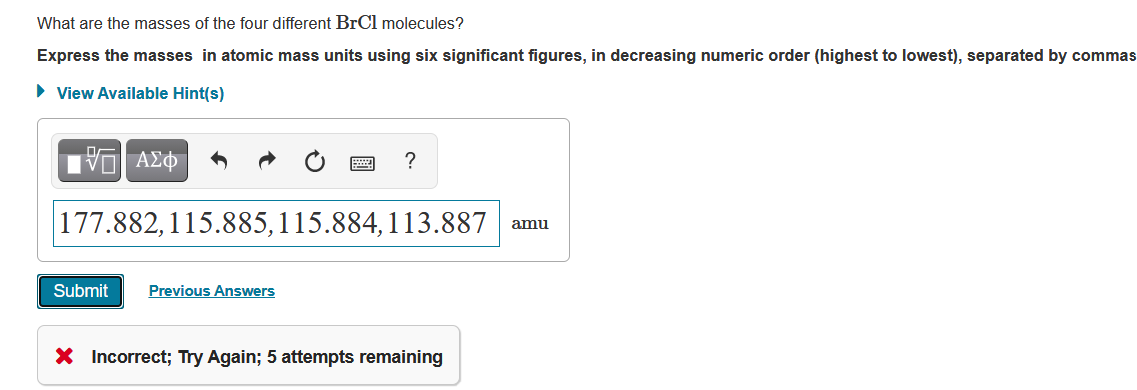
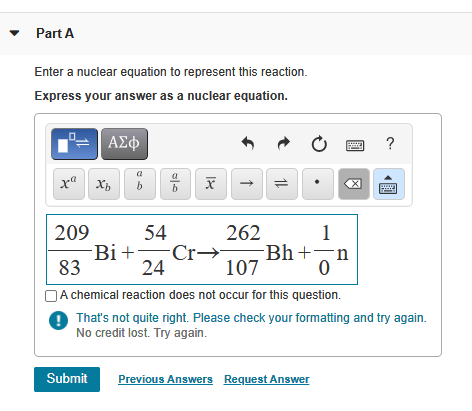
Transcribed Image Text:
What are the masses of the four different BrCl molecules? Express the masses in atomic mass units using six significant figures, in decreasing numeric order (highest to lowest), separated by commas ► View Available Hint(s) VE ΑΣΦ 177.882,115.885, 115.884,113.887 Submit Previous Answers ? X Incorrect; Try Again; 5 attempts remaining amu Part A Enter a nuclear equation to represent this reaction. Express your answer as a nuclear equation. O ΑΣΦ a xa Xb b Submit a b X Ĵ ↑ 14 Ć 209 54 262 1 -Bi+ Cr→ -Bh+n 24 83 107 0 A chemical reaction does not occur for this question. That's not quite right. Please check your formatting and try again. No credit lost. Try again. ? Previous Answers Request Answer What are the masses of the four different BrCl molecules? Express the masses in atomic mass units using six significant figures, in decreasing numeric order (highest to lowest), separated by commas ► View Available Hint(s) VE ΑΣΦ 177.882,115.885, 115.884,113.887 Submit Previous Answers ? X Incorrect; Try Again; 5 attempts remaining amu Part A Enter a nuclear equation to represent this reaction. Express your answer as a nuclear equation. O ΑΣΦ a xa Xb b Submit a b X Ĵ ↑ 14 Ć 209 54 262 1 -Bi+ Cr→ -Bh+n 24 83 107 0 A chemical reaction does not occur for this question. That's not quite right. Please check your formatting and try again. No credit lost. Try again. ? Previous Answers Request Answer
Expert Answer:
Answer rating: 100% (QA)
BrCl BROMINE CHLORIDE Based on the occurrence of different st... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For each compound below, what are the masses of the charged fragments produced by the indicated cleavage pathways? 7. 8. Refer to Exhibit 12-3. Alpha cleavage of Alpha cleavage and dehydration of OH
-
What are the masses and structures of the ions produced in the following cleavage pathways? (a) A-cleavage of 2-methyl-3-hexanone (two pathways) (b) Dehydration of cyclopentanol (c) McLafferty...
-
What are the masses of the charged fragments produced in the following cleavage pathways? (a) Alpha cleavage of 2-pentanone (CH 3 COCH 2 CH 2 CH 3 ) (b) Dehydration of Cyclohexanol (hydroxy...
-
Given that y = x 2 , find the image of the curve y = x 2 after each of the following combinations of transformations. a. A stretch in the y-direction with factor 3 followed by a translation by the...
-
The model for making tactical decisions described in the text has six steps. These steps are listed, out of order, below. Required: Put the steps in the correct order, starting with the step that...
-
In auction bid-ding, the "winner's curse" is the phenomenon of the winning (or highest) bid price being above the expected value of the item being auctioned. The Review of Economics and Statistics...
-
Explain the meaning of bond ratings and their impact on buying decisions.
-
The D.J. Masson Corporation needs to raise $500,000 for 1 year to supply working capital to a new store. Masson buys from its suppliers on terms of 3/10, net 90, and it currently pays on the 10th day...
-
In the SEC filing by the new management of Healthsouth, it was noted that the audit reports by Ernst & Young "did not contain an adverse opinion or disclaimer of opinion, nor were they qualified o...
-
Refer to Figure 7.16. Assume that the head loss in the pipe is given by h L = 0.02(L/D)(V 2 /2g), where V is the mean velocity in the pipe, D is the pipe diameter, and L is the pipe length. The...
-
This information relates to Windsor, Inc. for the year 2017. Retained earnings, January 1, 2017 $76,380 advertising expenses 2,052 dividends 6,840 Rental costs 11,856 Service revenues 66,120 Public...
-
How much Will it cost to print a circular sign with a radius of 1 . 6 feet if the printing company charges $ 2 9 per square foot? Use A = nr 2 , where is approximately 3 . 1 4 . ( round your answer...
-
Twenty gallons of pure water is in a tank. Water that contains 08 pounds of salt per gallon is pumped into the tank at a rate of 3 gallons per minute. Assuming the water is kept thoroughly mixed, how...
-
The objective of the assignment is exposing the students to new sustainable technologies ( products , processes , software, etc. ) and the Life Cycle Analysis of Such technologies. The students will...
-
Given: The state of stress at a point is represented by the element shown below. Required: a) Draw Mohr's circle. b) Calculate the principal stresses using the stress transformation equations. c)...
-
Lear is to become a partner in the WS partnership by paying $80,000 in cash to the business. At present, the capital balance for Hamlet is $70,000 and for MacBeth is $40,000. Hamlet and MacBeth share...
-
Hello please can someone help me to qnwer this Assessment of the CSR reports of SHISEIDO, how they match with actual actions/assessments available, including carbon footprint, water footprint and any...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
A point source emits radiation in all directions at a rate of 7.5 kW. What is the intensity of the radiation 5.0 m from the source?
-
A mass is suspended by two cords as shown in Fig. 8.44. What are the tensions in the cords? cord 1 45 1.5 kg
-
(a) What is the de Broglie wavelength of the Earth in its orbit about the Sun? (b) Treating the Earth as a de Broglie wave in a large gravitational atom, what would be the principal quantum number,...
-
Contractors Ltd was formed on 1 January 19X6 and the following purchases and sales of machinery were made during the first 3 years of operations. Each machine was estimated to last 10 years and to...
-
The financial year of H Saunders ended on 31 December 19X6. Show the ledger accounts for the following items including the balance transferred to the necessary part of the final accounts, also the...
-
On 1 January 19X8 the following balances, among others, stood in the books of M Baldock, a sole trader: (a) Rates, 104 (Dr); (b) Packing materials, 629 (Dr). During the year ended 31 December 19X8...

Study smarter with the SolutionInn App


