What is the difference in mass Am, at room temperature (T = 300 K) and atmospheric...
Fantastic news! We've Found the answer you've been seeking!
Question:
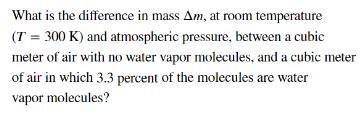
Transcribed Image Text:
What is the difference in mass Am, at room temperature (T = 300 K) and atmospheric pressure, between a cubic meter of air with no water vapor molecules, and a cubic meter of air in which 3.3 percent of the molecules are water vapor molecules? What is the difference in mass Am, at room temperature (T = 300 K) and atmospheric pressure, between a cubic meter of air with no water vapor molecules, and a cubic meter of air in which 3.3 percent of the molecules are water vapor molecules?
Expert Answer:
Answer rating: 100% (QA)
To calculate the difference in mass between dry air and air with water vapor you can use the ideal g... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, ? Ronald W. Rousseau, ? Lisa G. Bullard
Posted Date:
Students also viewed these physics questions
-
George is an avid skier and plans to spend each of the first four weekends in January skiing. However, because he is applying for jobs, he needs to skip one skiing weekend to update his resume....
-
What is the mass of a cubic meter of air at room temperature (20C)?
-
A flask with a volume of 1.50 L, provided with a stopcock, contains ethane gas (C2H6) at 300 K and atmospheric pressure (1.013 X l05 Pa). The molar mass of ethane is 30.1 g/mol. The system is warmed...
-
Generate a matrix of random integer temperatures in Fahrenheit from 70 to 100 for 10 weeks (rows) and 7 days per week (columns). The result should look something like this. Assume the first column is...
-
Walton Plastics manufactures custom park furniture and signage from recycled plastics (primarily shredded milk jugs.) Many of the company's customers are municipalities that are required by law to...
-
The following information for Great Oaks Furniture, a retail furniture and design firm, relates to Exercises 6 through 11. Calculate asset turnover, accounts receivable turnover, days sales in...
-
The payroll software is date sensitive, and it depends on the payroll transaction dates to accurately accumulate and process payroll data. (True/False)
-
The SEC Form 10-K of Google can be found online at www.wiley.com/college/pratt. REQUIRED: Review the Google SEC From 10-K, and answer the following questions: a. Does Google carry investment...
-
1. Find the solution set of each equation in the interval: 0 x <2". 4. cos 2x = 2 A. {111} (66 5 7 11 6 6 6 6 C. 3 3 3 D. T 5 3'3
-
Stan Barker opened Quik-Stop Market on January 3, 2017. The business is subject to FICA taxes. At the end of the first quarter of 2017, Barker, as president of the company, must file Form 941,...
-
The reaction of triphenyl methyl chloride (trityl) (A) and methanol (B) was carried out in a solution of benzene and pyridine at 25 C. (C,Hs),CCI + CH,OH > (C,H5);COCH, + HCI + D A C Pyridine reacts...
-
On January 1, 2024, Splash City issues $310,000 of 9% bonds, due in 20 years, with interest payable semiannually on June 30 and December 31 each year. Assuming the market interest rate on the issue...
-
Taveras Corporation is currently operating at 5 0 % of its available manufacturing capacity. It uses a job - order costing system with a plantwide predetermined overhead rate based on machine -...
-
Huron Company produces a commercial cleaning compound known as Zoom. The direct materials and direct labor standards for one unit of Zoom are given below: Standard Quantity or Hours Standard Price or...
-
Cordova manufactures three types of stained glass windows, cleverly named Products A, B, and C. Information about these products follows: Product A Product B Product C $ Sales price $51.00 $ 61.00...
-
Ashton Company, a distributor of exercise equipment, is preparing a cash budget for December. It provided the following information: a. The cash balance on December 1 is $50,600. b. Actual sales for...
-
The petty cash fund of Bridgeport's Auto Repair Service, a sole proprietorship, contains the following. 1. Coins and currency 2. Postage stamps 3. An I.O.U. from Richie Cunningham, an employee, for...
-
What is an insurable interest? Why is it important?
-
A stream of oxygen at 65C and 8.3 atm flows at a rate of 250 kg/h. Use the SRK equation of state to estimate the volumetric flow rate of this stream.
-
Air in industrial plants is subject to contamination by many different chemicals, and companies must monitor ambient levels of hazardous species to be sure they are below limits specified by the...
-
The wastewater treatment plant at the Ossabaw Paper Company paper mill generates about 24 tonnes of sludge per day. The consistency of the sludge is 35%, meaning that the sludge contains 35 wt%...
-
The Welding Department of Nagano Manufacturing Company has the following production and manufacturing cost data for February 2002. All materials are added at the beginning of the process. Nagano uses...
-
With the popularity of the Apple iPad, the tablet computer market is experiencing high growth rates and record demand levels. Using the Internet as a research tool, complete a five forces analysis...
-
The ledger of Grogan Company has the following work in process account. Production records show that there were 800 units in the beginning inventory, 30% complete, 1,100 units started, and 1,300...

Study smarter with the SolutionInn App


