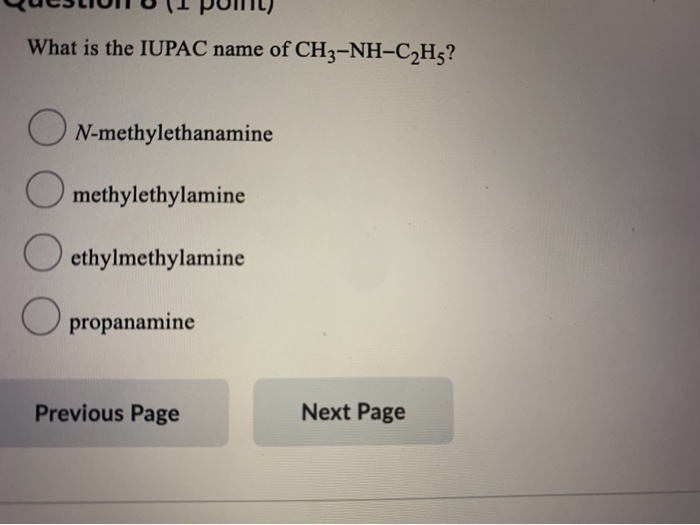
What is the IUPAC name of CH3-NH-CH5? ON-methylethanamine methylethylamine ethylmethylamine propanamine Previous Page Next Page If...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
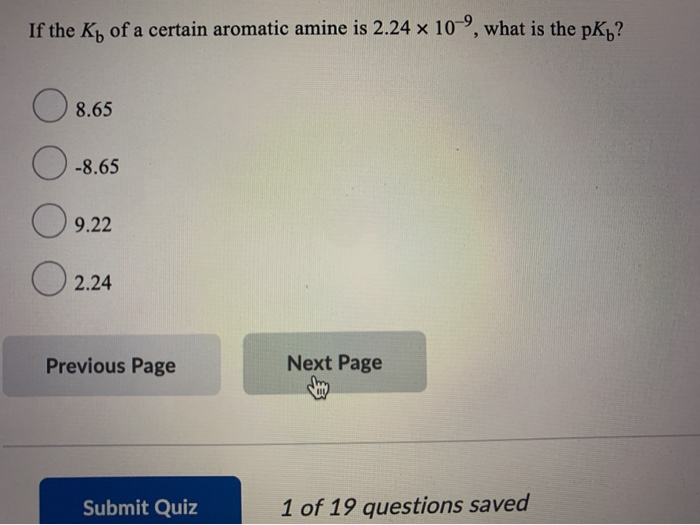
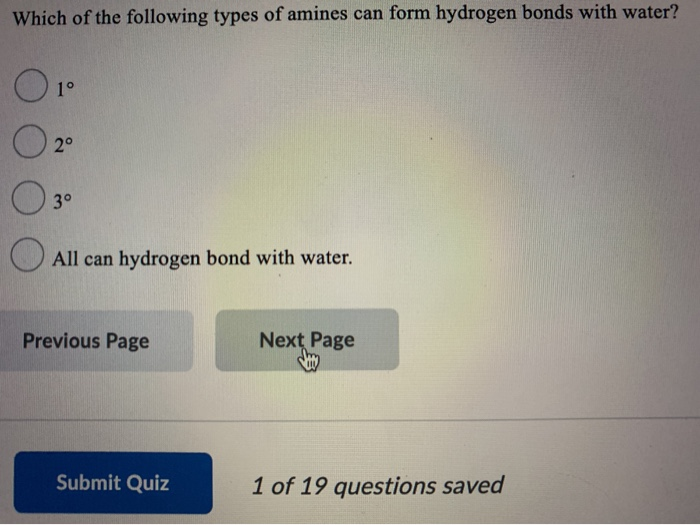
What is the IUPAC name of CH3-NH-C₂H5? ON-methylethanamine methylethylamine ethylmethylamine propanamine Previous Page Next Page If the Kt of a certain aromatic amine is 2.24 x 10, what is the pK₁? 8.65 -8.65 9.22 2.24 Previous Page Submit Quiz Next Page 1 of 19 questions saved Which of the following types of amines can form hydrogen bonds with water? 0 1° O 2⁰ 3° All can hydrogen bond with water. Previous Page Submit Quiz Next Page 1 of 19 questions saved What is the IUPAC name of CH3-NH-C₂H5? ON-methylethanamine methylethylamine ethylmethylamine propanamine Previous Page Next Page If the Kt of a certain aromatic amine is 2.24 x 10, what is the pK₁? 8.65 -8.65 9.22 2.24 Previous Page Submit Quiz Next Page 1 of 19 questions saved Which of the following types of amines can form hydrogen bonds with water? 0 1° O 2⁰ 3° All can hydrogen bond with water. Previous Page Submit Quiz Next Page 1 of 19 questions saved
Expert Answer:
Answer rating: 100% (QA)
Solution The polynomial is going through three data points tv v and t3v3 where from the above table ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the IUPAC name of this polymer? I-U-I I-C-H I-C-H I-U-H I-C-H I-C-H I-C-H I-U-H
-
What is the IUPAC name of the compound shown? (a) (E )-3-Methyl-2-hexenoic acid (b) (Z )-3-Methyl-2-hexenoic acid (c) (E )-3-Methyl-3-hexenoic acid (d) (Z )-3-Methyl-3-hexenoic acid H;C CO.H C=C...
-
What is the IUPAC name of the compound shown in the margin? (a) Isopropyl 2-fluoro3-methylbutanoate; (b) 2-fluoroisobutanoyl 2-propanoate; (c) 1-methylethyl 2-fluorobutyrate; (d) 2-fluoroisopropyl...
-
Under what circumstance is it most appropriate to travel in reverse with a load? A. When you are carrying a non-standard load B. When a tall load obstructs your forward vision C. When the load is...
-
In 1944, Benjamin Feinberg bought a plot in Beth Israel Cemetery in Plattsburgh, New York. A mausoleum was built on the plot to contain six crypts. In 1954, Feinbergs spouse died and was interred in...
-
The United States was on a gold standard from 1879 to 1914. During that period, the average inflation rate was about zero. Inflation was sometimes positive and sometimes negative, and there was no...
-
\(12+(13-6) \times 8-130\) Perform the indicated calculation.
-
Property Tax Calculations and Journal Entries. The Village of Darbys budget calls for property tax revenues for the fiscal year ending December 31, 2011, of $2,660,000. Village records indicate that,...
-
I need help responding to this. Globalization and becoming a global citizen have its benefits as well as the negative side of it. When it comes to international drug trafficking, the implications...
-
Smoothie Plc, a fruit juice manufacturer, has determined the following standard cost details for the three ingredients that are blended together to make their tropical fruit drink: Standard cost per...
-
When goods are shipped FOB shipping point, the purchaser will increase Inventory and typically decreases Cash for the freight cost under a perpetual system. True False
-
You are an equity trader in an over-leveraged hedge fund. Your manager urgently needs your analysis of the cost of liquidating a very large block of shares in Boeing with immediate effect. She wants...
-
The police were called to Latheef Street where a woman had been robbed. When they arrived, the robber had gone. Apart from the victim, there were three witnesses to the robbery, who each gave the...
-
Bert & Ernie are running a race; when the starting pistol fires, Bert sprints forward with a constant speed of 7 m.s; Ernie is still tying his shoes when the pistol fires and begins running 6 seconds...
-
How many times do you need to deallocate memory (using either delete[] or delete) to prevent this code from leaking? float myData[3]; float myPtr nullptr; for (int i = 0; i < 2; i++) myData[i] new...
-
What does a welfare and progressive tax system do to the effects of luck and distribution of disposable income and happiness ? Explain the potential positive and negative effects of a welfare...
-
Do you plan to publish your research? No Why or why not? Unable to spend the time needed to collect data. Plan to focus on finding a position in Human Resources or I/O psychology. What might be some...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
(a) Predict the direction of reaction when chlorine gas is added to an equilibrium mixture of COCl 2 , CO, and Cl 2 . The reaction is COCl 2 (g) CO(g) + Cl 2 (g) (b) What is the direction of...
-
The n quantum number of an atomic orbital is 6. What are the possible values of l? What are the possible values of ml if the l quantum number is 5?
-
Consider the complex ion [CoF6]3. a. What is the geometry? b. Which is a more likely color for this ion to absorb, red or blue? c. Would you expect this complex to be high or low spin?
-
Write out the full accounting equation.
-
State two different ways in which profit can be measured.
-
In the absence of any new capital introduced, or any withdrawal of capital by the owner, what will cause the balance on the owner's equity account to change from year to year?

Study smarter with the SolutionInn App


