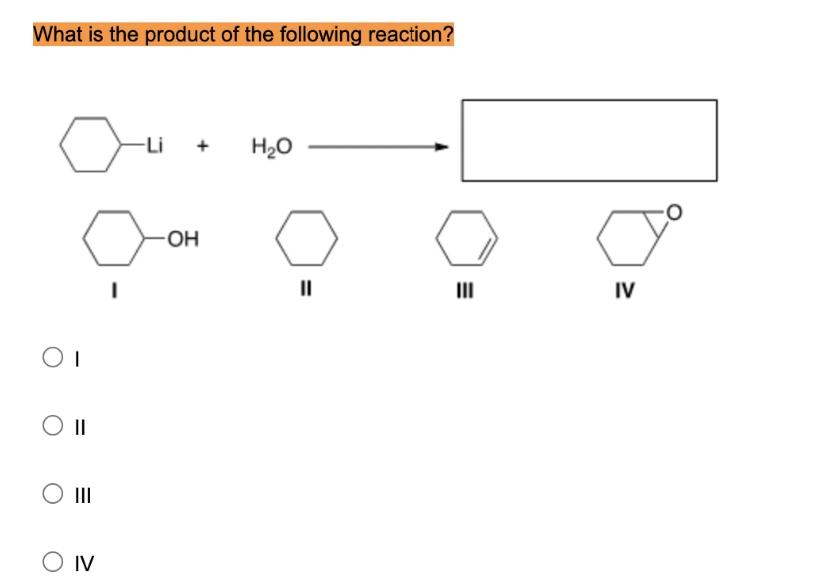
What is the product of the following reaction? OI -OH O Q O II O III...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
What is the product of the following reaction? OI -OH O Q O II O III -Li O IV H₂O IV What is the product of the following reaction? OI -OH O Q O II O III -Li O IV H₂O IV
Expert Answer:
Answer rating: 100% (QA)
II cyclohexane Here is the handwritten explanation wit... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
What is the product of the following SuzukiMiyaura coupling? Br B(OR)2 + Pd catalyst Base
-
What is the product of the following reaction? A) I B) II C) III D) IV NH2 (2 equivalents) I =c-c-CH;CH, H H =c-C-CHCH, HH CEC-CH,CH C=C-CH,CH, IV
-
What is the product of the following reaction? A) I B) II C) III D) IV NH2 (2 equivalents) I =c-c-CH;CH, H H =c-C-CHCH, HH CEC-CH,CH C=C-CH,CH, IV
-
Consider the no-trade input/output situation presented in the following table for countries X and Y. Assuming that free trade is allowed, develop a scenario that will benefit the citizens of...
-
What is job evaluation? Explain the differences between the major job evaluation systems, noting the advantages and disadvantages of each.
-
The manager of the local Walmart Supercenter is studying the number of items purchased by customers in the evening hours. Listed below is the number of items for a sample of 30 customers. a. Find the...
-
Answer the same question as in problem 3 but for the simple implicit method. problem 3 Consider the heat equation (6.1) with \(a=0.1\) solved in the interval \(0
-
The Village of Budekville, which has a fiscal year July 1 to June 30, sold $3,000,000 in 6 percent tax-supported bonds at par to construct an addition to its police station. The bonds were dated and...
-
Question 1. Cash Flows (2 pts.) Use the following information for the problems (assume the tax rate is 34 percent): 2014 2015 Sales $9,402 $10,091 Depreciation 1,350 1,351 Cost of goods sold 3,235...
-
Roger Ginde is developing a program in supply chain management certification for managers. Ginde has listed a number of activities that must be completed before a training program of this nature...
-
a) Prove that if p and q are odd primes and q dividesa p -1, then either q divides a-1 or q = 2kp + 1 for someinteger k. b) Use part a to show that if p is an odd prime, then theprime divisors of 2 p...
-
The Dougherty Furniture Company manufactures tables. In March, the production department had budgeted allocation base (i.e. cost driver) of 4,000 machine-hours. The budgeted manufacturing overheads...
-
Body Images Pty Ltd (BI) is a beauty therapy company in Perth, Western Australia. They sell various beauty services to clients who want to improve their personal image. BI operates out of a square...
-
Let A = 3 Find B = sin(A) Find C = A Find D=C2 Find ECC Find F = C.*C % Your script goes here.
-
A company applies overhead at a rate of 160% of direct labor cost. Actual overhead cost for the current period is $1,006,000, and direct labor cost is $590,000. Determine whether there is over- or...
-
Fanning Industries produces two electronic decoders, P and Q. Decoder P is more sophisticated and requires more programming and testing than does Decoder Q. Because of these product differences, the...
-
The following lots of Commodity Z were available for sale during the year. Beginning inventory 11 units at $47 First purchase Second purchase Third purchase 19 units at $50 54 units at $57 17 units...
-
d) For die casting processes: 1. What are the most common metals processed using die casting and discuss why other metals are not commonly die casted? 2. Which die casting machines usually have a...
-
Which would you expect to be the stronger acid? Explain your reasoning in each instance. (a) CH2ClCO2H or CHCl2CO2H (b) CCl3CO2H or CHCl2CO2H (c) CH2FCO2H or CH2BrCO2H (d) CH2FCO2H or CH2FCH2CO2H
-
When, in separate reactions, 2-methylpropene, propene, and ethene are allowed to react with HI under the same conditions (i.e., identical concentration and temperature), 2-methylpropene is found to...
-
(a) Show how you might use a cyclic acetal in carrying out the following transformation: (b) Why would a direct addition of methylmagnesium bromide to A fail to give B? OH
-
The company had one temporary difference due to the GAAP basis of equipment exceeding the tax basis of the equipment. Record the income tax journal entry for 2020, assuming an enacted tax rate of...
-
Evergreen Company's 2020 reconciliation between pretax GAAP income and taxable income is as follows. Pretax GAAP income.. Depreciation adjustment. Permanent difference. Taxable income.. $ 200,000...
-
Lake Company has pretax GAAP income of \(\$ 100,000\) in 2020, its first year of operations. Lake Company has depreciation expense in 2020 for GAAP purposes that is \(\$ 60,000\) less than the amount...

Study smarter with the SolutionInn App


