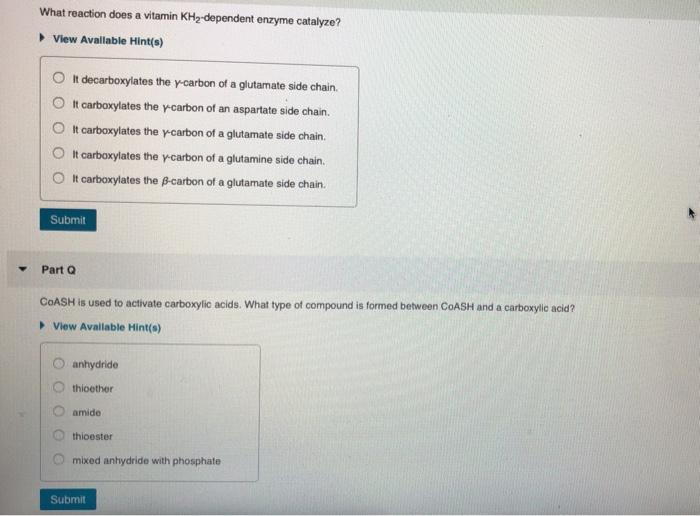
What reaction does a vitamin KH-dependent enzyme catalyze? View Available Hint(s) O It decarboxylates the y-carbon...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
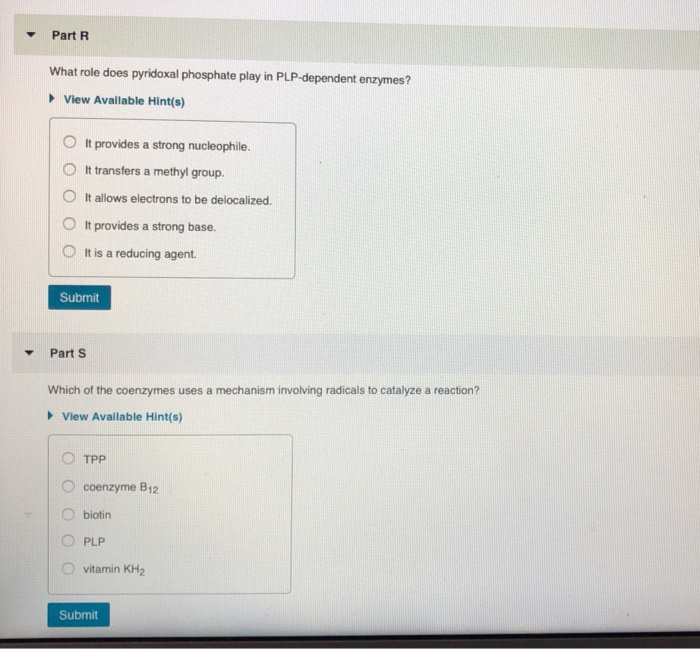
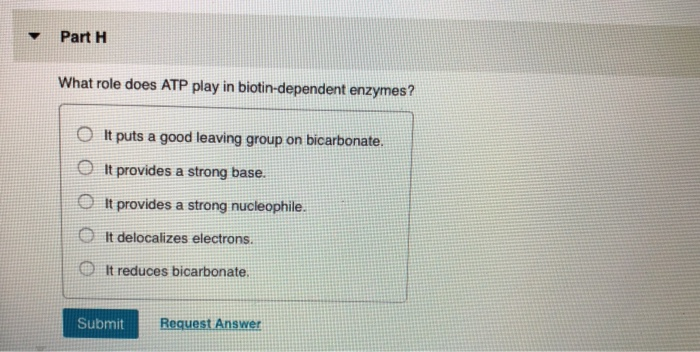
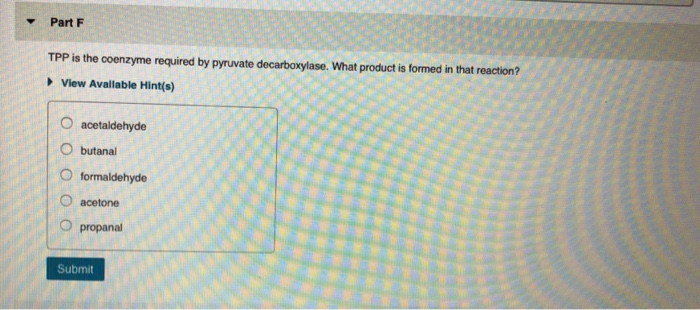
▼ What reaction does a vitamin KH₂-dependent enzyme catalyze? ►View Available Hint(s) O It decarboxylates the y-carbon of a glutamate side chain. OIt carboxylates the y-carbon of an aspartate side chain. O It carboxylates the y-carbon of a glutamate side chain. OIt carboxylates the y-carbon of a glutamine side chain. O It carboxylates the ß-carbon of a glutamate side chain. Submit Part Q COASH is used to activate carboxylic acids. What type of compound is formed between COASH and a carboxylic acid? View Available Hint(s) anhydride thioether amide thioester mixed anhydride with phosphate Submit ▼ Part R What role does pyridoxal phosphate play in PLP-dependent enzymes? ▸ View Available Hint(s) O It provides a strong nucleophile. OIt transfers a methyl group. It allows electrons to be delocalized. OIt provides a strong base. O It is a reducing agent. Submit Part S Which of the coenzymes uses a mechanism involving radicals to catalyze a reaction? ▸ View Available Hint(s) TPP coenzyme B12 biotin PLP vitamin KH₂ Submit Part T What is the first step in the mechanism for the conversion of uracil to thymine catalyzed by thymidylate synthase using NN0-methylenetetrahydrofolate as a coenzyme? View Available Hint(s) nucleophilic attack on the B-carbon of uracil by a nucleophilic cysteine of the enzyme O deprotonation of uracil O nucleophilic attack on the B-carbon of uracil by a nucleophilic nitrogen of the enzyme nucleophilic attack by uracil on 5.10-methylenetetrahydrofolate nucleophilic attack by a nucleophilic cysteine residue of the enzyme on the carbonyl carbon of uracil OO O Submit ▼ Part H What role does ATP play in biotin-dependent enzymes? It puts a good leaving group on bicarbonate. OIt provides a strong base. It provides a strong nucleophile. It delocalizes electrons. It reduces bicarbonate. Submit Request Answer ▼ Part F TPP is the coenzyme required by pyruvate decarboxylase. What product is formed in that reaction? ► View Available Hint(s) O acetaldehyde O butanali formaldehyde O acetone O propanal Submit ▼ What reaction does a vitamin KH₂-dependent enzyme catalyze? ►View Available Hint(s) O It decarboxylates the y-carbon of a glutamate side chain. OIt carboxylates the y-carbon of an aspartate side chain. O It carboxylates the y-carbon of a glutamate side chain. OIt carboxylates the y-carbon of a glutamine side chain. O It carboxylates the ß-carbon of a glutamate side chain. Submit Part Q COASH is used to activate carboxylic acids. What type of compound is formed between COASH and a carboxylic acid? View Available Hint(s) anhydride thioether amide thioester mixed anhydride with phosphate Submit ▼ Part R What role does pyridoxal phosphate play in PLP-dependent enzymes? ▸ View Available Hint(s) O It provides a strong nucleophile. OIt transfers a methyl group. It allows electrons to be delocalized. OIt provides a strong base. O It is a reducing agent. Submit Part S Which of the coenzymes uses a mechanism involving radicals to catalyze a reaction? ▸ View Available Hint(s) TPP coenzyme B12 biotin PLP vitamin KH₂ Submit Part T What is the first step in the mechanism for the conversion of uracil to thymine catalyzed by thymidylate synthase using NN0-methylenetetrahydrofolate as a coenzyme? View Available Hint(s) nucleophilic attack on the B-carbon of uracil by a nucleophilic cysteine of the enzyme O deprotonation of uracil O nucleophilic attack on the B-carbon of uracil by a nucleophilic nitrogen of the enzyme nucleophilic attack by uracil on 5.10-methylenetetrahydrofolate nucleophilic attack by a nucleophilic cysteine residue of the enzyme on the carbonyl carbon of uracil OO O Submit ▼ Part H What role does ATP play in biotin-dependent enzymes? It puts a good leaving group on bicarbonate. OIt provides a strong base. It provides a strong nucleophile. It delocalizes electrons. It reduces bicarbonate. Submit Request Answer ▼ Part F TPP is the coenzyme required by pyruvate decarboxylase. What product is formed in that reaction? ► View Available Hint(s) O acetaldehyde O butanali formaldehyde O acetone O propanal Submit
Expert Answer:
Answer rating: 100% (QA)
part 1 question ans Vitamin Kdependent enzymes play an important role in the posttranslational modifications required for various physiological processes including blood coagulation and bone mineraliz... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In glycine, the carboxylic acid group has Ka = 4.3 X 10-3 and the amino group has Kb = 6.0 X 10-5. Use these equilibrium constant values to calculate the equilibrium constants for the following. a....
-
Vitamin B6 (an aldehyde) reacts with an enzyme (partial structure shown below) to form a coenzyme that catalyzes the conversion of α-amino acids (Chapter 17) to α-keto...
-
A carboxylic acid is formed when an α haloketone reacts with hydroxide ion. This reaction is called a Favorskii reaction. Propose a mechanism for the following Favorskii reaction....
-
On March 31, 2019, the balances of the accounts appearing in the ledger of Racine Furnishings Company, a furniture wholesaler, are as follows: a. Prepare a multiple-step income statement for the year...
-
Create a process flowchart for an activity that you do dailyfor example, getting ready for school each day.
-
A crate on a motorized cart starts from rest and moves with a constant eastward acceleration of = 2.80 m/s 2 . A worker assists the cart by pushing on the crate with a force that is eastward and has...
-
The balance sheet of Creative Communications, Inc., reported the following: Preferred dividends are in arrears for three years including the current year. On the balance sheet date, the market value...
-
1. Why are antidumping (AD) and countervailing duty (CVD) measures brought and imposed? 2. What is the impact on a firm from a nontriad country if it faces an AD or CVD case in its major market?
-
Assume you are trying to estimate the price you will pay for a bond. The face value is $1000, the coupon rate of interest paid annually is 10%, the bonds mature in 25 years and are currently priced...
-
Watershed is a media services company that provides online streaming movie and television content. As a result of the competitive market of streaming service providers, Watershed is interested in...
-
13.8 Let f be holomorphic in C. (i) Prove that if |f(z)| > M in C then f is constant. 160 Cauchy's formulae (ii) Prove that if e is bounded then f is constant.
-
Suppose a seven-year $1,000 bond with a 7.04% coupon rate and semiannual coupons is trading with a yield to maturity of 4.87% a. Is this bond currently trading at a discount, at par, or at a premuim?...
-
For a 0.66 specific gravity gas at 250 F, calculate and plot pseudopressures in a pressure range from 14.7 psia and 8,000 psia. Under what condition is the pseudopressure linearly proportional to...
-
A company has to produce 100,000 units of output. It has to choose among three policies i.e. policy A, B & C. Policy A proposes to maintain current assets of worth 10 lacs, policy B of 8 lacs and...
-
A loan of $87,100 is due 10 years from today. The borrower wants to make annual payments at the end of each year into a sinking that will earn compound interest at an annual rate of 10 percent....
-
The $45.0 million lottery payment that you have just won actually pays $3.0 million per year for 15 years. The interest rate is 9%. a. If the first payment comes in 1 year, what is the present value...
-
Consider the 1st order reversible reaction AB, where the rate constant for the forward reaction (A goes to B) is k, and the rate constant for the reverse reaction (B goes to A) is k. Imagine that...
-
XYZ Inc. a calendar year, accrual basis corporation, had the following items during 2021: Gross revenue from operations Cost of goods sold $420,000 ($180,000) $9,000 LT capital gain .LT capital...
-
Write a structure for each of the following compounds: a. isopropyl alcohol b. isopentyl fluoride c. sec-butyl iodide d. neopentyl chloride e. tert-butylamine f. n-octyl bromide
-
Which of the following pairs of dinucleotides are present in equal amounts in DNA? a. CC and GG b. CG and GT c. CA and TG d. CG and AT e. GT and CA f. TA and AT
-
Write the mechanism for the base-catalyzed conversion of D-fructose into D-glucose and D-mannose.
-
Following is a probability density curve with the area between 0 and 1 and the area between 1 and 2 indicated. a. What proportion of the population is between 0 and 1? b. What is the probability that...
-
Use Table A.2 to find the area between z = 1.13 and z = 2.02. Z 0.00 0.01 0.02 0.03 0.04 0.05 0.06 0.07 0.08 0.09 -0.8 -0.7 .2119 .2090 .2420 2389 -0.6 .2743 .2709 .2676 .2061 .2033 2358 .2327 .2643...
-
Use Table A.2 to find the area to the left of z = 0.25. Z 0.00 0.01 0.02 0.03 0.04 0.05 0.06 0.07 0.08 0.09 -0.8 -0.7 .2119 .2090 .2420 2389 -0.6 .2743 .2709 .2676 .2061 .2033 2358 .2327 .2643 .2005...

Study smarter with the SolutionInn App


