When aqueous solutions of barium nitrate and ammonium sulfate are mixed, the balance equation for this...
Fantastic news! We've Found the answer you've been seeking!
Question:

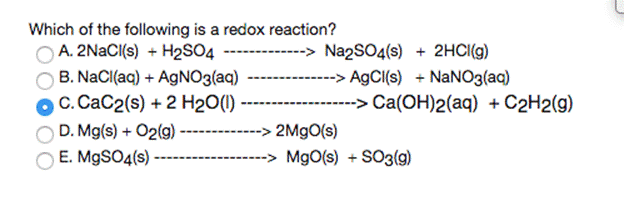

Transcribed Image Text:
When aqueous solutions of barium nitrate and ammonium sulfate are mixed, the balance equation for this reaction is: Ba(NO3)2 (aq) + (NH4)2SO4 (aq)------ - 2 NH4NO3 (aq) + BaSO4(s) What are the "spectator ions" in the reaction? O Ba2+ and NO3" O NH4* and NO3 O so42- and NO3¯ NH4+ and SO42- O Ba2+ and SO42- Which of the following is a redox reaction? A. 2NACI{s) + H2S04 ---- B. NaCl(aq) + AGNO3(aq) OC. CaC2(s) + 2 H2O(1) D. Mg(s) + O2(g) E. M9SO4(s) -> Na2SO4(s) + 2HCI(g) --> A9CI(s) + NaNO3(aq) --> Ca(OH)2(aq) + C2H2(g) 2M9O(s) -> MgO(s) + SO3(9) 2.5000 g of KNO3 is dissolved in 200.0 ml aqueous solution that has density of 1.034 g/mL. Calculate for KNO3 in the solution: Do not write down the answers in exponential form molality 0.0022 When aqueous solutions of barium nitrate and ammonium sulfate are mixed, the balance equation for this reaction is: Ba(NO3)2 (aq) + (NH4)2SO4 (aq)------ - 2 NH4NO3 (aq) + BaSO4(s) What are the "spectator ions" in the reaction? O Ba2+ and NO3" O NH4* and NO3 O so42- and NO3¯ NH4+ and SO42- O Ba2+ and SO42- Which of the following is a redox reaction? A. 2NACI{s) + H2S04 ---- B. NaCl(aq) + AGNO3(aq) OC. CaC2(s) + 2 H2O(1) D. Mg(s) + O2(g) E. M9SO4(s) -> Na2SO4(s) + 2HCI(g) --> A9CI(s) + NaNO3(aq) --> Ca(OH)2(aq) + C2H2(g) 2M9O(s) -> MgO(s) + SO3(9) 2.5000 g of KNO3 is dissolved in 200.0 ml aqueous solution that has density of 1.034 g/mL. Calculate for KNO3 in the solution: Do not write down the answers in exponential form molality 0.0022
Expert Answer:
Answer rating: 100% (QA)
Q1 spectator ions will not form any preciptiate form the list NO3 is on bo... View the full answer

Related Book For 

Fundamentals of biochemistry Life at the Molecular Level
ISBN: 978-0470547847
4th edition
Authors: Donald Voet, Judith G. Voet, Charlotte W. Pratt
Posted Date:
Students also viewed these accounting questions
-
What are the spectator ions in the reaction between aqueous hydrobromic acid and aqueous sodium hydroxide? H + and OH Na + only H + , Br , Na + , and OH Na + and Br Br only
-
Aldolase catalyzes the reaction ÎG°² for this reaction is 22.8 kJmol-1. In the cell at 37°C, the ÎG for this reaction is -5.9 kJmol-1. What is the ratio [GAP][DHAP]/[FBP]?...
-
What are the spectator ions present when aqueous solutions of titanium(IlI) acetate and sodium chromate are mixed? Include charges, and use superscripts and/or subscripts if necessary (click on the...
-
What is the discount yield, bond equivalent yield, and effective annual return on a $ 5 million commercial paper issue that currently sells at 98.625 percent of its face value and is 136 days from...
-
Primorsk Corporation began the year 2011 with cash of $27,950. In addition to earning a net income of $19,000 and paying a cash dividend of $9,750, Primorsk borrowed $39,000 from the bank and...
-
1. Is Summit Software a gazelle? Support your answer. 2. What problems may Jim face owning such a fast-growing business? 3. Are gazelles more important to the economy than traditional growth...
-
Label each of the following characteristics of a corporation as either an (A) advantage or a (D) disadvantage: a. Organizational costs b. Continuity of existence c. Capital raising capability d....
-
Even though most corporate bonds in the United States make coupon payments semiannually, bonds issued elsewhere often have annual coupon payments. Suppose a German company issues a bond with a par...
-
2/174 The vertical shaft of the industrial robot rotates at the constant rate w. The length h of the verti- cal shaft has a known time history, and this is true of its time derivatives and as well....
-
The smooth rod of mass M rests inside the glass. Determine the reactions on the rod. Given: M = 20 gm a = 75 mm b = 200 mm = 40 deg g = 9.81m/s2 b.
-
List and describe the dimensions of Non Verbal Communication. Provide an example of how one culture differs from another on each dimension. 9. What is the global youth culture. What evidence do you...
-
Modifying behavior is when the manager focuses on Research and compare behavior assessment instruments available on the market today. Select and evaluate two behavior assessment tools by completing a...
-
How do cultural policies and interventions, enacted by state institutions or grassroots movements, influence cultural production, dissemination, and participation, and shape notions of cultural...
-
The plant is not operating at full capacity. Presently, the volume of production is 248,769 units. The fixed overhead cost is P189,535 under the present operating conditions. The costs per unit are...
-
What ethical considerations arise when engaging in cross-cultural research and representation, particularly in terms of cultural appropriation, representation, and the ethics of cultural exchange ?
-
City Bicycle, a cycling store has a beginning inventory of one bi-level touring bicycle 1, for which it paid $2,500. Suppose that during the period the store purchases bi-level touring bicycle 2 for...
-
! Required information The rotating shaft in the given figure is machined from AISI 1020 CD steel. It is subjected to a force of F= 8 kN. Find the minimum factor of safety for fatigue based on...
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
In what way is UMP synthesis similar to fatty acid synthesis in mammals?
-
How does the binding of ligand to a receptor tyrosine kinase lead to changes in gene expression?
-
Below is a beaker that contains a solution of 0.1 M NaCl. Floating inside is a cellulose bag that is permeable to water and small ions like Na+ and Cl- but is impermeable to protein. The bag contains...
-
What is rational ignorance?
-
The ability-to-pay principle states: a. Those with the greatest ability to pay taxes should pay more. b. Those with the least ability to pay taxes should pay more. c. Individuals receiving the...
-
What is the median voter model?

Study smarter with the SolutionInn App


