When determining the heat capacity of a coffee cup calorimeter, Sammy uses the same volumes and...
Fantastic news! We've Found the answer you've been seeking!
Question:
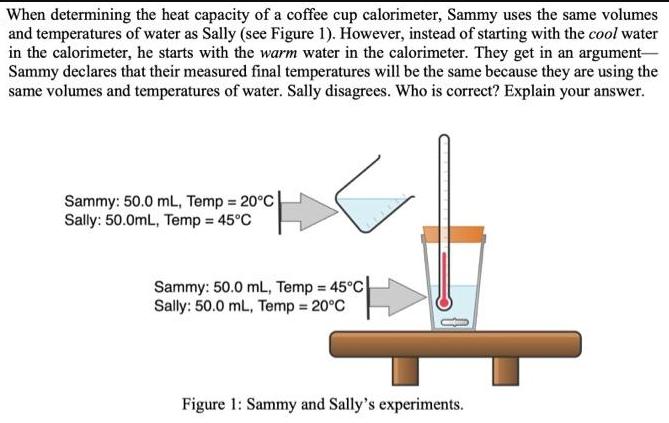
Transcribed Image Text:
When determining the heat capacity of a coffee cup calorimeter, Sammy uses the same volumes and temperatures of water as Sally (see Figure 1). However, instead of starting with the cool water in the calorimeter, he starts with the warm water in the calorimeter. They get in an argument- Sammy declares that their measured final temperatures will be the same because they are using the same volumes and temperatures of water. Sally disagrees. Who is correct? Explain your answer. Sammy: 50.0 mL, Temp = 20°C Sally: 50.0mL, Temp = 45°C Sammy: 50.0 mL, Temp = 45°C Sally: 50.0 mL, Temp = 20°C Figure 1: Sammy and Sally's experiments. When determining the heat capacity of a coffee cup calorimeter, Sammy uses the same volumes and temperatures of water as Sally (see Figure 1). However, instead of starting with the cool water in the calorimeter, he starts with the warm water in the calorimeter. They get in an argument- Sammy declares that their measured final temperatures will be the same because they are using the same volumes and temperatures of water. Sally disagrees. Who is correct? Explain your answer. Sammy: 50.0 mL, Temp = 20°C Sally: 50.0mL, Temp = 45°C Sammy: 50.0 mL, Temp = 45°C Sally: 50.0 mL, Temp = 20°C Figure 1: Sammy and Sally's experiments.
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below Sally is correct While both Sammy and S... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
1. What is the present value (PV") of an offer of $15,000 two years from now if the Opportunity Cost of Capital (OCC) is $12% per annum? 2. What is the PV of an offer to receive $12,000 three years...
-
The budget director of Birds of a Feather Inc., with the assistance of the controller, treasurer, production manager, and sales manager, has gathered the following data for use in developing the...
-
What are the pros and cons of using the Six Sigma approach?
-
Locate the income statement of a company in each of the following industries: pharmaceutical, technology, retailergroceries, and automobile manufacturer. Calculate the gross profit margin, operating...
-
On December 1, Marzion Electronics Ltd. has three DVD players left in stock. All areidentical all are priced to sell at $150. One of the three DVD players left in stock, withserial #1012, was...
-
The notion that "math is a universal language" implies that language minority students will have fewer problems with mathematics than with Language Arts is not supported by either research or...
-
You have just been promoted to manage the process defined by the five stages A to E below. After three months on the job you realize something is not right with process capacity because your...
-
Considering the financial system requirement, it was decided that company will use MYOB to handle company's finances. Write an email to the manager (trainer/assessor) requesting the equipment,...
-
Visit and review the website for Gopher Industries ( https://www.gopherresource.com ). Also watch the video link on its website about the lead recylcling process (...
-
Forgotten Worlds is a project about representation. Compose an piece that exposes a current or ongoing underrepresented issue, event, or topic that is not being covered or represented very well in...
-
18. Qualified Small Business Stock. (Obj. 1) Jenson pays $30,000 for Section 1202 qualified small business stock on February 16, 2005. In October 2018, Jenson sells the stock for $80,000. a. Compute...
-
Analyze the genetic basis of neurodegenerative disorders, focusing on the role of protein misfolding, aggregation, and proteostasis dysfunction in conditions such as Alzheimer's and Parkinson's...
-
1. If Indian River Citrus, does not have the opportunity to lease the space, does this mean that space is free or costless, from the standpoint of the lite product project? 2. Should the erosion of...
-
Suppose that a flow network G = (V, E) violates the assumption that the network contains a path s t for all vertices V. Let u be a vertex for which there is no path s u t. Show that there must...
-
How would your answers change if the temperature was increased by 50C, the particle diameter was doubled, and fluid velocity was cut in half? Assume properties of water can be used for this system.
-
Make up and solve an original problem. The guidelines are given in Problem VO do Ub UR VO
-
1. What is the NFPA diamond and why is it important? 2. Go to Wikipedia and search ethylene oxide to find the NFPA diamond numbers. 3. Draw the NFPA diamond for the chemicals propylene, isohexane,...
-
The figure below illustrates a thin plate of thickness \(t\). An approximate displacement field, which accounts for displacements due to the weight of the plate, is given by a. Determine the...
-
The stress matrix at a particular point in a body is Determine the corresponding strain if \(E=20 \times 10^{10} \mathrm{~Pa}\) and \(v=0.3\). -2 1-3 [0] = 104 -345 107 Pa.
-
For a plane stress problem, the strain components in the xyplane at a point \(P\) are computed as: a. Compute the state of stress at this point if Young's modulus \(E=2 \times 10^{11} \mathrm{~Pa}\)...

Study smarter with the SolutionInn App


