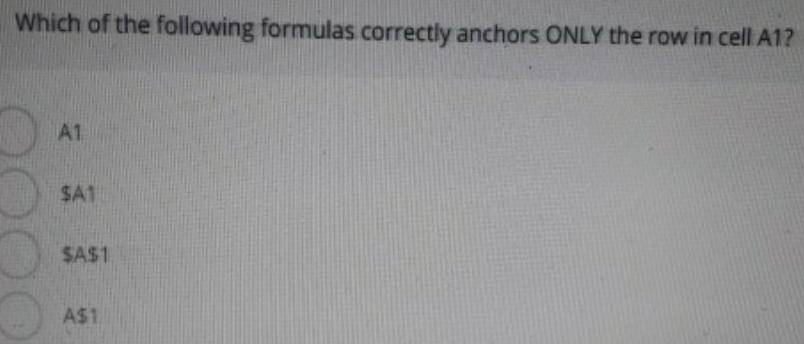
Which of the following formulas correctly anchors ONLY the row in cell A1? A1 SA1 SAS1...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Which of the following formulas correctly anchors ONLY the row in cell A1? A1 SA1 SAS1 A$1 Which of the following formulas correctly anchors ONLY the row in cell A1? A1 SA1 SAS1 A$1
Expert Answer:
Answer rating: 100% (QA)
Answer A1 Explanation An absolute reference in Excel is ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
One of the manual transmission operations costs the wages of workers 4200 dinars annually, and it was found that the client can be performed using automated transport equipment that costs workers'...
-
Which of the following formulas for (f, g) define inner products on the space C°[ - 1, 1 ]? a. b. c. d. Fee f(x)g(x)e dx f(x) g(x)x dx f (x)g(x) (x + 2) dx f(x)g(x)x2 dx
-
Which of the following formulas define norms on R3? (a) ||v|| = 2v21 + v22 + 3v23 (b) ||v|| = v21 + 2v1v2 + v22 + v23 (c) ||v|| = max {v1,|, |v2|, |v3|} (d) ||v|| = {v1, - v2| + |v2 - v3| + |v3 - v1}...
-
On December 31, 2021, there is a batch of commodities sold under FOB destination conditions in the shipping area, and this batch of commodities is not included in the inventory count middle. There is...
-
The U.S. Federal Highway Administration conducts studies on motor vehicle travel by type of vehicle. Results are published annually in Highway Statistics. A sample of 15 cars yields the following...
-
There is associated with each atom in paramagnetic and ferromagnetic materials a net magnetic moment. Explain why ferromagnetic materials can be permanently magnetized whereas paramagnetic ones...
-
Source Today earned net income of \(\$ 60,000\) after deducting depreciation of \(\$ 4,000\) and all other expenses. Current assets decreased by \(\$ 3,000\), and current liabilities increased by...
-
Compute the contribution margin ratio and the breakeven point in total sales dollars for Wall Street Products if the selling price per unit is $16, the variable cost per unit is $8, and the fixed...
-
This unit discussed civil laws. Reflect on the term liability as an emergency services officer. What types of liability could you or your agency face in the scope of its duties?
-
At December 31, 2010, Westport Manufacturing Co. owned the following investments in the capital stock of publicly owned companies (all classified as available-for-sale securities): In 2011, Westport...
-
4] Delete the root twice from this heap. (First 100, then whatever the next root will be.) 100 (17) 19 3 36 25 1 2 7 Redraw the heap after each change and explain what has happened. What is the total...
-
Your original post should include at least one reference to an article, paper, or other online material from a reputable source on one of the topics listed below (or an original topic of your...
-
The following information represents the cash transactions of Mr. Favour for the month of September 2020: 2020 September 1 Balances brought forward: Cash RM2,870; Bank RM21,426. 2 The following paid...
-
Pintoo industries Ltd is considering the replacement of one of its moulding machines. The existing machine is in good operating condition, but is smaller than required if the firm is to expand its...
-
Wildhorse Corporation was organized on January 1, 2019. During its first year, the corporation issued 1,950 shares of $50 par value preferred stock and 104,000 shares of $10 par value common stock....
-
An electric utility is considering a new power plant in northern Arizona. Power from the plant would be sold in the Phoenix area, where it is badly needed. Because the firm has received a permit, the...
-
Beginning inventory, purchases, and sales for Item 88-HX are as follows: Jan. 1 Inventory 110 units @ $32 8 Sale 88 units 15 Purchase 122 units @ $34 27 Sale 102 units Assuming a perpetual inventory...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Can you construct a linear function L: R3 R such that and If yes, find one. If not, explain why not. 011
-
(a) Show that if v is a linear combination of v1.......vm, and each vj is a linear combination of w1,..., wm then v is a linear combination of w1..........wn. (b) Suppose v1.....vm, span V. Let...
-
The formulas serve to define the basic complex trigonometric functions. Write out the formulas for their real and imaginary parts in terms of z = x + i y. and show that cos z and sin z reduce to...
-
What is the IQR? What does the IQR represent? The population parameters below describe the full-time equivalent number of students (FTES) each year at Lake Tahoe Community College from 19761977...
-
How many standard deviations away from the mean is the median? The population FTES for 20052006 through 20102011 was given in an updated report. The data are reported here. The population parameters...
-
A class of 20 students has a mean grade of 80 on a test. Nineteen of the students has a mean grade between 79 and 82, inclusive. a. What is the lowest possible grade of the other student? b. What is...

Study smarter with the SolutionInn App


