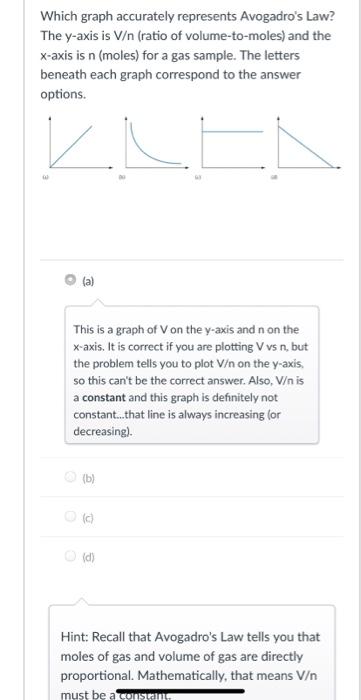
Which graph accurately represents Avogadro's Law? The y-axis is V/n (ratio of volume-to-moles) and the x-axis...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
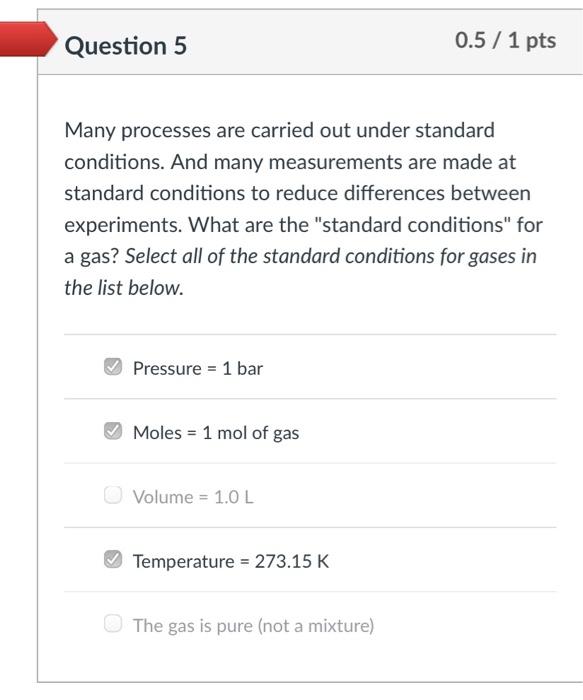
Which graph accurately represents Avogadro's Law? The y-axis is V/n (ratio of volume-to-moles) and the x-axis is n (moles) for a gas sample. The letters beneath each graph correspond to the answer options. KLEN This is a graph of V on the y-axis and n on the x-axis. It is correct if you are plotting V vs n, but the problem tells you to plot V/n on the y-axis, so this can't be the correct answer. Also, V/n is a constant and this graph is definitely not constant.that line is always increasing (or decreasing). O (b) O (c) O (d) Hint: Recall that Avogadro's Law tells you that moles of gas and volume of gas are directly proportional. Mathematically, that means V/n must be aconstant Question 5 0.5 /1 pts Many processes are carried out under standard conditions. And many measurements are made at standard conditions to reduce differences between experiments. What are the "standard conditions" for a gas? Select all of the standard conditions for gases in the list below. Pressure = 1 bar Moles = 1 mol of gas Volume = 1.0 L Temperature = 273.15 K The gas is pure (not a mixture) Which graph accurately represents Avogadro's Law? The y-axis is V/n (ratio of volume-to-moles) and the x-axis is n (moles) for a gas sample. The letters beneath each graph correspond to the answer options. KLEN This is a graph of V on the y-axis and n on the x-axis. It is correct if you are plotting V vs n, but the problem tells you to plot V/n on the y-axis, so this can't be the correct answer. Also, V/n is a constant and this graph is definitely not constant.that line is always increasing (or decreasing). O (b) O (c) O (d) Hint: Recall that Avogadro's Law tells you that moles of gas and volume of gas are directly proportional. Mathematically, that means V/n must be aconstant Question 5 0.5 /1 pts Many processes are carried out under standard conditions. And many measurements are made at standard conditions to reduce differences between experiments. What are the "standard conditions" for a gas? Select all of the standard conditions for gases in the list below. Pressure = 1 bar Moles = 1 mol of gas Volume = 1.0 L Temperature = 273.15 K The gas is pure (not a mixture)
Expert Answer:
Answer rating: 100% (QA)
Question 4 Answer c According to Avogadros law V n V volume n moles Vn constant Volume by m... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A tank volume V contains gas at conditions (0, p0, T0). At time t = 0 it is punctured by a small hole of area A. According to the theory of Chap. 9, the mass flow out of such a hole is approximately...
-
Under what conditions is it important to send business letters rather than e-mail messages?
-
You are already plotting that to answer Romans burning questions by your March 15 deadline, you need to evaluate the software as well as the vendors, systematically, and then render a decision....
-
There are different types of non-probability sampling methods. Below are examples of such methods. You are required to go through them and (for each example); a. Name the Sample Method b. Write a not...
-
The Maruska Corporation has outstanding $800,000 of 8 percent bonds callable at 104. On September 1, immediately after recording the payment of the semiannual interest and the amortization of the...
-
On January 1, 2014, Doone Corporation acquired 60 percent of the outstanding voting stock of Rockne Company for $300,000 consideration. At the acquisition date, the fair value of the 40 percent...
-
The 2014 financial statements of LVMH Moet Hennessey-Louis Vuitton S.A. are presented in Appendix C of this book. LVMH is a Paris-based holding company and one of the worlds largest and best-known...
-
Maria Lorenzi owns an ice cream stand that she operates during the summer months in West Yellowstone, Montana. Her store caters primarily to tourists passing through town on their way to Yellowstone...
-
What does your overall profile suggest in relation to your needs for personal and professional development? Based on the competency most in need of development, identify three possible actions that...
-
In this mini-case, you will complete the test of details on accounts receivable for the 2019 audit of EarthWear Clothiers, Inc. The principal test of detail involves sending "confirmations" or...
-
3. Find the indefinite integral: 4. Find the indefinite integral: 5. Find the indefinite integral: 2014 (4x-4) dx (3 marks) 2x6 (x7 +1) 6 dx (3 marks) 196rx 3xx 9e dx (3 marks)
-
Examine the influence of oral traditions on contemporary written forms of storytelling, such as in works of Indigenous and African diaspora literature. How do these traditions shape the structure and...
-
Analyze the role of narrative voice in the development of detective fiction. How does the use of first-person and third-person narration alter the reader's experience and engagement with the mystery?
-
Pretty Paws Salon is the only small business in town to provide pet grooming services. The table shows the demand schedule for the Pretty Kitty service package (bathing, ear cleaning, and nail...
-
Every investment carries the expectation of a return, which is represented as the additional percentage rate that we expect to obtain when investing our money in a financial instrument. It is...
-
Corporation, Inc. has the following income statement (in millions) (2pts.): A1 CORPORATION, INC. Income Statement For the Year Ended December 31, 2026 Net Sales Cost of Goods Sold Gross Profit...
-
2. Use the tombstone notation to illustrate the following workflow. Describe the steps for translating a C->Python program written in C, on an X86 processor. Draw the Tombstone notation to show how...
-
What are three disadvantages of using the direct write-off method?
-
The aorta carries blood away from the heart at a speed of about 40 cm/s and has a radius of approximately 1.1 cm. The aorta branches eventually into a large number of tiny capillaries that distribute...
-
The biologically equivalent dose for a typical chest X-ray is 2.5 10-2 rem. The mass of the exposed tissue is 21 kg, and it absorbs 6.2 10-3 J of energy. What is the relative biological...
-
Along a straight road through town, there are three speed-limit signs. They occur in the following order: 55, 35, and 25 mi/h, with the 35-mi/h sign located midway between the other two. Obeying...
-
PhotoMax, Inc., plans to prepare a sales budget schedule and a purchases budget schedule for 20X3. Management has set a sales goal of 250,000 units. After reviewing price trends, the sales manager...
-
Repeat Problem 15.D27, but bulb 2 at \(\mathrm{z}=\delta\) contains \(\mathrm{y}_{\mathrm{air}}=0.610, \mathrm{y}_{\mathrm{H} 2}=0.010\), and \(\mathrm{y}_{\mathrm{NH} 3}=0.380\). Problem 15.D27 This...
-
On December 31, 20X2, the accounting records of Zylar, Inc., show the following unit sales for 20X2. The following are additional actual amounts for the 4th quarter of 20X2. Management has...

Study smarter with the SolutionInn App


