Which is the correct order of reagents you would need to use for the following synthesis?...
Fantastic news! We've Found the answer you've been seeking!
Question:
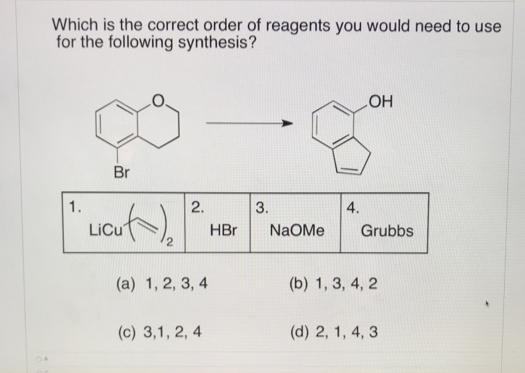
Transcribed Image Text:
Which is the correct order of reagents you would need to use for the following synthesis? HO Br 1. 2. 3. 4. LiCu HBr NaOMe Grubbs (a) 1, 2, 3, 4 (b) 1, 3, 4, 2 (c) 3,1, 2, 4 (d) 2, 1, 4, 3 Which is the correct order of reagents you would need to use for the following synthesis? HO Br 1. 2. 3. 4. LiCu HBr NaOMe Grubbs (a) 1, 2, 3, 4 (b) 1, 3, 4, 2 (c) 3,1, 2, 4 (d) 2, 1, 4, 3
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Determine CPI, ETC (1), and EAC. Activity Total PV 1 2 3 4 5 6 7 8 9 10 11 12 13 14 1 6 6 2 20 10 10 3 30 9 6 6 6 3 4 20 8 2 5 5 5 16 4 4 4 4 6 18 9 9 7 8 4 4 Status Report: Ending Period 5 Task %...
-
Allergan is a major pharmaceutical firm. You work for Allergan's CFO and are evaluating a major and expensive drug trial. The drug trial would require an investment of $95 million today If the trial...
-
a. Make an exponential smoothing forecast for periods 2 through 5 with 2 values of alpha, 0.05 and 0.60, and an assumed forecast for period 1 of 30. b. Compute the MAD for each of the above...
-
Examine how effective change communication can reduce resistance in organization ? explain
-
Two recent business school graduates, Jane Johns and Lou Schwartz, started a shop called Exports Unlimited on January 1. Their partnership agreement stipulated that each would receive 10 percent on...
-
Suppose k, l, and m grow at constant rates given by k, l, and m. What is the growth rate of y in each of the following cases? (a) y = k1/3 (b) y = k1/3l2/3 (c) y = mk1/3l2/3 (d) y = mk1/4l3/4 (e) y =...
-
Graph the expectation function for the logistic growth model (12.34) for \(\theta_{1}=10, \theta_{2}=2\), and values of \(\theta_{3}=0.25,1,2,3\), respectively. Overlay these plots on the same set of...
-
Following is a list of cost system characteristics and sample companies. Match each to either job order costing or process costing. a. Companies that produce small quantities of many different...
-
What is the MOS of a 14-20 bolt holding the beam below to the wall O O O O O Assume beam is 6061-T6, as is the wall. Assume the CoF is 1.5 Assume the bolt hole is oversized by 0.030" Assume the bolts...
-
Decision analyst Sandy Baron has taken a job with an up-and-coming consulting firm in San Francisco. As part of the move, Sandy will purchase a house in the area. There are two houses that are...
-
Why attorneys are reluctant to provide auditors with unlimited statements about contingencies?
-
Consider the DE = 2x(2000-x). This is a logistic DE. (a) If a(t) is a solution to this DE then what are the possible values for limitt-x(t). (b) If a solution r(t) to this DE also satisfies 0 < x(0)...
-
-5 a) Calculate 2 = (-23+2i) and write the answer Cartesian form. on b) solve the equation 2+(4+2i)z+ (12-4) 0 +H
-
Another model of population growth, say for cells of a tumor, is the differential equation for p(t) > 0. (a) The population is increasing p'(t) = 2p(t)in (p(t)) (b) The population is decreasing when...
-
a.) Prove that if A 0 and B 0, then AB 0, assuming that the product AB is defined. b.) (5 points) Prove that if A 0 and B 0, then AB 0, assuming that the product AB is defined. c.) (5 points)...
-
Recall that we considered the vector space of real-valued, continuous functions on [0, 1], de- noted by C[0, 1]. Equip this space with the standard inner product, with (f, g) = f f(t)g(t)dt. Also...
-
2) Silver price today is $14 per ounce; the futures price for November 2019 is $14.5 per ounce. There are 5,000 ounces of silver in each futures contract. The margin requirement for each contract is...
-
Consider a game of poker being played with a standard 52-card deck (four suits, each of which has 13 different denominations of cards). At a certain point in the game, six cards have been exposed. Of...
-
Consider the following melting point data: Account for the trends in melting points for the two series of compounds in terms of interparticle forces. Compound NaCI MgCl AICl SiC PCl SC2 CIh mp (C)...
-
Consider the general reaction aA + bB cC and the following average rate data over a specific time period t: A/t = 0.0080 mol L-1s-1 B/t = 0.0120 mol L-1s-1 C/t = 0.0160 mol L-1s-1 Determine a set...
-
You are studying the kinetics of the reaction H2(g) + F2(g) 2HF(g) and you wish to determine a mechanism for the reaction. You run the reaction twice by keeping one reactant at a much higher pressure...
-
A 1.0-cm-diameter sphere is charged to a potential of \(3400 \mathrm{~V}\). How much charge is on the sphere?
-
a. What is the potential difference between the terminals of an ordinary AA or AAA battery? (If you're not sure, find one and look at the label.) b. An AA battery is connected to a parallel-plate...
-
\(\mathrm{A}+25 \mathrm{nC}\) charge is at the origin. How much farther from the charge is the \(2000 \mathrm{~V}\) equipotential surface than the \(3000 \mathrm{~V}\) surface?

Study smarter with the SolutionInn App



