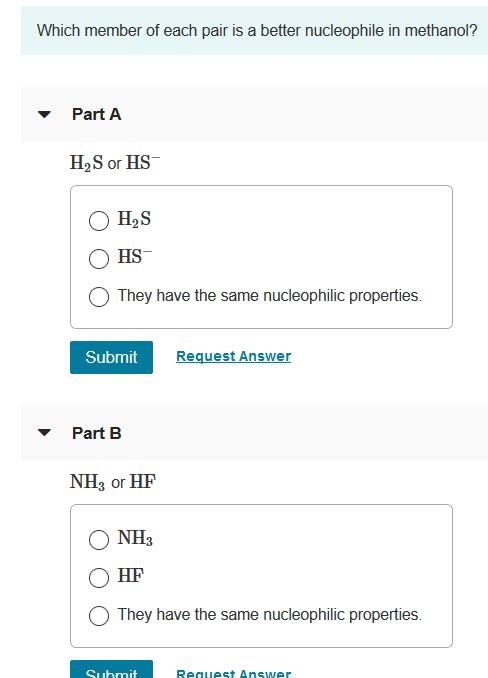
Which member of each pair is a better nucleophile in methanol? Part A H2S or HS...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
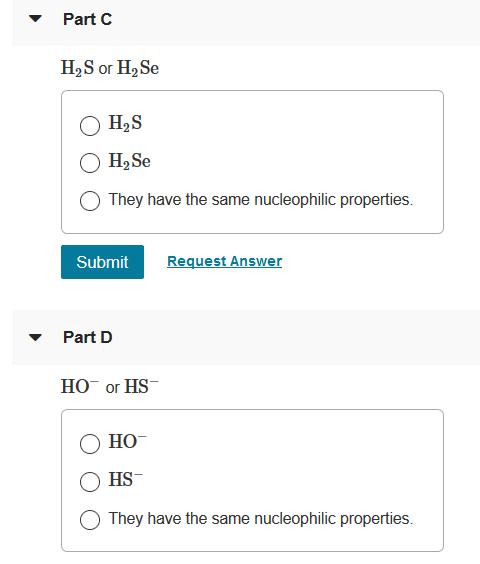
Which member of each pair is a better nucleophile in methanol? Part A H2S or HS H2S HS- They have the same nucleophilic properties. Submit Request Answer Part B NH3 or HF NH3 HF They have the same nucleophilic properties. Submit Request Answer Part C H,S or H2 Se H,S H2Se They have the same nucleophilic properties. Submit Request Answer Part D НО ог HS НО HS- They have the same nucleophilic properties. Part E I- or Cl- CI O They have the same nucleophilic properties. Submit Request Answer Part F Br or I- Br They have the same nucleophilic properties. Submit Request Answer Which member of each pair is a better nucleophile in methanol? Part A H2S or HS H2S HS- They have the same nucleophilic properties. Submit Request Answer Part B NH3 or HF NH3 HF They have the same nucleophilic properties. Submit Request Answer Part C H,S or H2 Se H,S H2Se They have the same nucleophilic properties. Submit Request Answer Part D НО ог HS НО HS- They have the same nucleophilic properties. Part E I- or Cl- CI O They have the same nucleophilic properties. Submit Request Answer Part F Br or I- Br They have the same nucleophilic properties. Submit Request Answer
Expert Answer:
Answer rating: 100% (QA)
Negative charge apecies are better nucleophile than reuteaf of any mole... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Which species in each pair is a better oxidizing agent under standard-state conditions? (a) Br2 or Au3+. (b) H2 or Ag+. (c) Cd2+ or Cr3+. (d) O2 in acidic media or O2 in basic media.
-
Which species in each pair is a better reducing agent under standard-state conditions? (a) Na or Li. (b) H2 or I2. (c) Fe2+ or Ag. (d) Br- or Co2+.
-
Predict which member of each pair is more acidic, and explain the reasons for your predictions. (a) Cyclopentanol or 3-chlorophenol (b) Cyclohexanol or cyclohexanethiol (c) Cyclohexanol or...
-
Below are three independent and unrelated errors. a. On December 31, 2020, Wolfe-Bache Corporation failed to accrue salaries expense of $1,800. In January 2021, when it paid employees for the...
-
Refer to Exercise 61. Which, if any, of the following factors would force you to change your answers? For each of the factors listed, discuss your reasoning. (Consider each part independent of the...
-
Draw a breakeven chart to illustrate an increase in advertising expenses.
-
How can social media aid in developing a questionnaire?
-
Consider the situation of La Nacin, a hypothetical Latin American country. In 2010, La Nacin was a net debtor to the rest of the world. Assume that all of La Nacins foreign debt was dollar...
-
[111] The inverse of the matrix A = 6 7 8 8 using 6 7-8 adjoint method is :
-
Capstone Case: Sunrise Bakery Expansion The Sunrise Bakery Corporation was originally founded in Houston, TX in 1991 by Griffin Harris, who currently serves as the company's Chief Executive Officer....
-
Use the ALEKS graphing calculator to find the: x-intercept(s) and vertex for the quadratic function. =fx-4x2-6x2 Round to the nearest hundredth if necessary. If there is more than one x-intercept,...
-
Assuming that the elastic bands stretch and store energy like a spring, how high would the \(80 \mathrm{~kg}\) pogo and rider go for a stretch of \(0.20 \mathrm{~m}\) ? A. \(2.0 \mathrm{~m}\) B....
-
Riders can use fewer elastic bands, reducing the effective spring constant of the pogo. The maximum stretch of the bands is still \(0.40 \mathrm{~m}\). Reducing the number of bands will A. Reduce the...
-
Explain the meaning of the following statement: "The probability that the discrete random variable takes the value 2 is 0.3 ."
-
Appreciate the wide range of nonlinear functions that can be estimated using a model that is linear in the parameters.
-
Explain how to compute a prediction, a prediction interval, and a goodness-of-fit measure in a log-linear model.
-
Darcy's law states that v=ki. This simple law is generally valid for seepage in soils of particle size up to coarse sands only, why is this so? [4 marks] A natural residual soil has the grain size...
-
Does log 81 (2401) = log 3 (7)? Verify the claim algebraically.
-
Roughly speaking, the radius of an atom is about 10,000 times greater than that of its nucleus. If an atom were magnified so that the radius of its nucleus became 2.0 cm, about the size of a marble,...
-
Why do the 3s, 3p, and 3d orbitals have the same energy in a hydrogen atom but different energies in a many-electron atom?
-
The formation of H2 from two H atoms is an energetically favorable process. Yet statistically there is less than a 100 percent chance that any two H atoms will undergo the reaction. Apart from energy...
-
Twitter is trading at \($34.50.\) Call options with a strike price of \($35\) are priced at \($2.30\) . What is the intrinsic value of the option, and what is the time value?
-
Name five variables that can affect the price of options, and briefly explain how each affects prices. How important are intrinsic value and time value to in-the-money options? To out-of-the-money...
-
Consider a put with a strike price of \($20\) and a premium of \($4.\) If the stock price is currently \($18,\) what is the maximum loss to the naked writer of the put? a. \($16\) b. $20 c. Unlimited

Study smarter with the SolutionInn App


