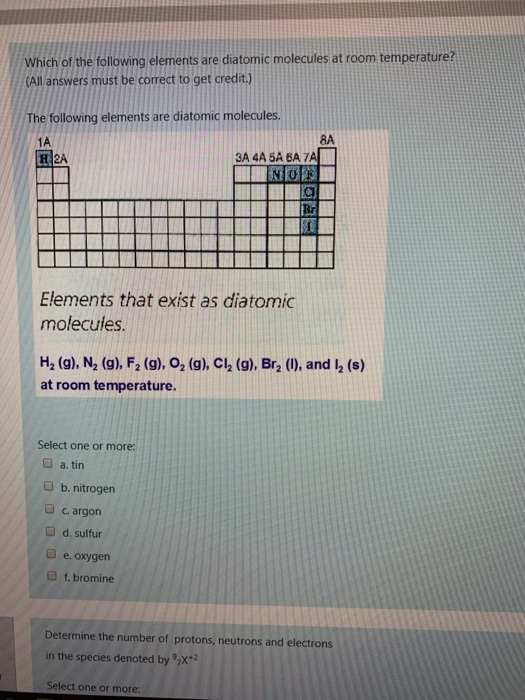
Which of the following elements are diatomic molecules at room temperature? (All answers must be correct...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
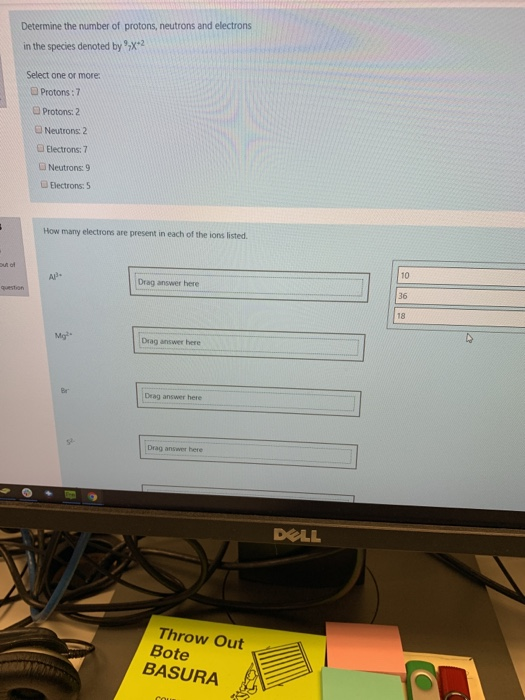
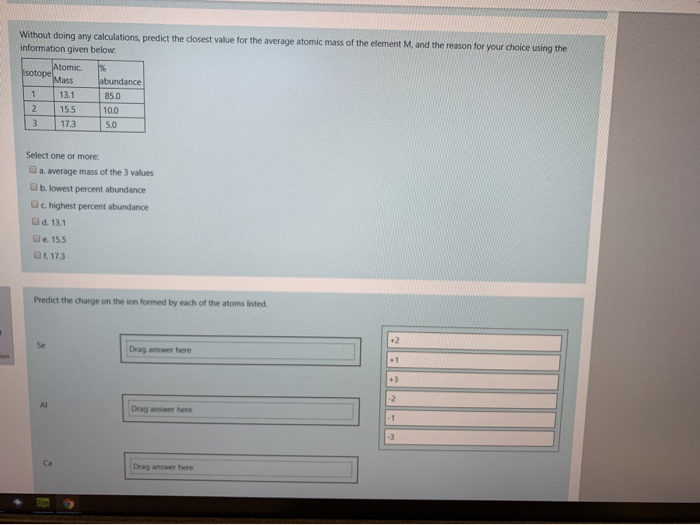
Which of the following elements are diatomic molecules at room temperature? (All answers must be correct to get credit.) The following elements are diatomic molecules. 1A H2A Elements that exist as diatomic molecules. Select one or more: a. tin b. nitrogen c. argon d. sulfur 3A 4A 5A 6A 7A NOF e. oxygen f. bromine H₂ (g), N₂ (g), F₂ (g), O₂ (g), Cl₂ (g), Br₂ (1), and I2₂ (s) at room temperature. C Br Select one or more: 8A Determine the number of protons, neutrons and electrons in the species denoted by X+2 S out of Determine the number of protons, neutrons and electrons in the species denoted by X-2 question Select one or more: Protons: 7 Protons: 2 Neutrons: 2 Electrons: 7 Neutrons: 9 Electrons: 5 How many electrons are present in each of the ions listed. Al- Mg Drag answer here Drag answer here Drag answer here Drag answer here Throw Out Bote BASURA COUR DELL 10 36 18 mon Without doing any calculations, predict the closest value for the average atomic mass of the element M, and the reason for your choice using the information given below. % sotope 1 2 3 Atomic. Mass 13.1 15.5 17.3 Select one or more: a. average mass of the 3 values b. lowest percent abundance c. highest percent abundance d. 13.1 e. 15.5 f. 17.3 Al 2 abundance 85.0 10.0 5.0 Predict the charge on the ion formed by each of the atoms listed. Ca Drag answer here Drag answer here Drag answer here +2 +1 +3 -2 -1 Which of the following elements are diatomic molecules at room temperature? (All answers must be correct to get credit.) The following elements are diatomic molecules. 1A H2A Elements that exist as diatomic molecules. Select one or more: a. tin b. nitrogen c. argon d. sulfur 3A 4A 5A 6A 7A NOF e. oxygen f. bromine H₂ (g), N₂ (g), F₂ (g), O₂ (g), Cl₂ (g), Br₂ (1), and I2₂ (s) at room temperature. C Br Select one or more: 8A Determine the number of protons, neutrons and electrons in the species denoted by X+2 S out of Determine the number of protons, neutrons and electrons in the species denoted by X-2 question Select one or more: Protons: 7 Protons: 2 Neutrons: 2 Electrons: 7 Neutrons: 9 Electrons: 5 How many electrons are present in each of the ions listed. Al- Mg Drag answer here Drag answer here Drag answer here Drag answer here Throw Out Bote BASURA COUR DELL 10 36 18 mon Without doing any calculations, predict the closest value for the average atomic mass of the element M, and the reason for your choice using the information given below. % sotope 1 2 3 Atomic. Mass 13.1 15.5 17.3 Select one or more: a. average mass of the 3 values b. lowest percent abundance c. highest percent abundance d. 13.1 e. 15.5 f. 17.3 Al 2 abundance 85.0 10.0 5.0 Predict the charge on the ion formed by each of the atoms listed. Ca Drag answer here Drag answer here Drag answer here +2 +1 +3 -2 -1
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following molecules may show a pure rotational microwave absorption spectrum? (a) H20, (b) H202, (c) NH4 (d) N20?
-
Which of the following molecules may show a pure rotational Raman spectrum? (a) CH2Cl2 (b) CH3CH3, (c) SP6, (d) N2O?
-
Which of the following molecules may show infrared absorption spectra? (a) CH3CH3, (b) CH4, (c) CH3CI, (d) N2
-
Miami Tropical stock has a beta of 1.25. The risk-free rate of return is 4.34 percent and the market risk premium is 9.02 percent. What is the expected rate of return on this stock? 10.23 percent...
-
Explain why, when all markets are competitive and in equilibrium, all three conditions for efficiency are satisfied. Does this result indicate that societys welfare is maximized?
-
A rod of 10-mm diameter and 250-mm length has one end maintained at 100C. The surface of the rod experiences free convection with the ambient air at 25C and a convection coefficient that depends on...
-
Maribel Baltazar was hired by clothing retail merchandiser Forever 21 in 2007. During the hiring process, Baltazar was given an 11-page document to sign, two pages of which contained an arbitration...
-
One day, while Bruno is preparing for a difficult stunt, he gets into an argument with the director and refuses to perform any stunts at all. Can X Entertainment seek specific performance of the...
-
The eigenvalues, eigenvectors in Question-1 needs to be calculated by hand (not by computer). Questions-1: (9 mark) A car rental company has three locations. Every customer can rent from and return...
-
The American company Green Plant Inc. (supplier) has signed a contract with the Argentinian company Agua Pura (buyer), to install a new bottling line in its old plant located in Buenos Aires. The...
-
Explain the relationship between liabilities and expenses. Provide five examples where they both increase at the same time (4 Marks). Why are some liabilities interest-bearing and others are not (3...
-
give a detailed explanation on every line (from book) Add a merge() method to the OrdArray class so that you can merge two ordered source arrays into an ordered destination array. Write code in...
-
Current Attempt in Progress On March 20, Monty's petty cash fund of $114 is replenished when the fund contains $12 in cash and receipts for postage $48, supplies $21, and travel expense $33. Prepare...
-
Sandhill Co., organized in 2024, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2025 and 2026. Intangible...
-
What is the main focus of incident reporting, occurrence reporting, and occurrence screening ?
-
7. Tucker Enterprises has a 12% cost of capital, and has generated the following information on two projects: Year Project 1 Project 2 012345 ($100,000) ($250,000) 30,000 75,000 30,000 75,000 30,000...
-
Consider a market where the market supply is given by the function Pg = 10 +1.5Qs and the market demand is given by Pp = 80-2QD- a. Find the market equilibrium price and quantity. b. Assume that due...
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
The radius of a uranium-235 nucleus is about 7.0 10-3 pm. Calculate the density of the nucleus in g/cm3. (Assume the atomic mass is 235 amu.)
-
Calculate the total number of electrons that can occupy (a) One s orbital, (b) Three p orbitals, (c) Five dorbitals, (d) Seven forbitals.
-
The fluorides of the second-period elements and their melting points are: LiF, 845C; BeF2, 800C; BF3, 2126.7C; CF4, -184C; NF3, 2206.6C; OF2, -223.8C; F2, -219.6C. Classify the type(s) of...
-
Prove the second part of Theorem 3.8. That is let \(\left\{X_{n}ight\}_{n=1}^{\infty}\) be a sequence of random variables, \(X\) be a random variable, and \(g\) be a Borel function on \(\mathbb{R}\)....
-
Prove the first part of Theorem 3.7. That is let \(\left\{X_{n}ight\}_{n=1}^{\infty}\) be a sequence of random variables, \(c\) be a real constant, and \(g\) be a Borel function on \(\mathbb{R}\)...
-
Let \(\left\{X_{n}ight\}_{n=1}^{\infty},\left\{Y_{n}ight\}_{n=1}^{\infty}\), and \(\left\{Z_{n}ight\}_{n=1}^{\infty}\) be independent sequences of random variables that converge in probability to the...

Study smarter with the SolutionInn App


