Calculate the mass in grams of aluminum iodide (All3) when 21.10 g of aluminum (Al) reacted...
Fantastic news! We've Found the answer you've been seeking!
Question:
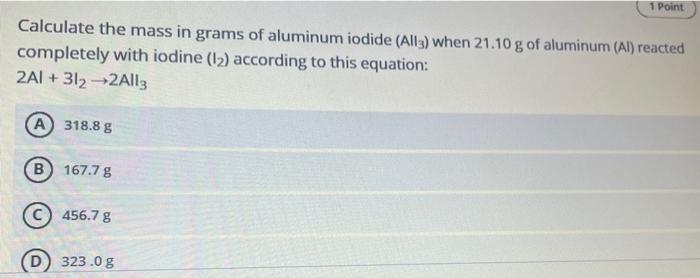
Transcribed Image Text:
Calculate the mass in grams of aluminum iodide (All3) when 21.10 g of aluminum (Al) reacted completely with iodine (1₂) according to this equation: 2AI +312-2All3 A) 318.8 g B) 167.7 g 456.7 g 1 Point 323.0 g Calculate the mass in grams of aluminum iodide (All3) when 21.10 g of aluminum (Al) reacted completely with iodine (1₂) according to this equation: 2AI +312-2All3 A) 318.8 g B) 167.7 g 456.7 g 1 Point 323.0 g Calculate the mass in grams of aluminum iodide (All3) when 21.10 g of aluminum (Al) reacted completely with iodine (1₂) according to this equation: 2AI +312-2All3 A) 318.8 g B) 167.7 g 456.7 g 1 Point 323.0 g Calculate the mass in grams of aluminum iodide (All3) when 21.10 g of aluminum (Al) reacted completely with iodine (1₂) according to this equation: 2AI +312-2All3 A) 318.8 g B) 167.7 g 456.7 g 1 Point 323.0 g
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below To solve this problem you can use the s... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
The Roman Empire lasted from 27 b.c. to a.d. 476. The empire was wealthy enough to build such monuments as the Roman Coliseum. Roman engineering skill was at a level high enough that aqueducts built...
-
S and B corporations are members of an affiliated group that has filed consolidated tax returns for several years. S drills a water well for B in Year 1 and charges B $5,000 for the service. S incurs...
-
After the fall of the Roman Empire, new Germanic kingdoms arose and became known as Latin Christendom blending Latin and Germanic traditions. Discuss examples of these traditions and how they evolved...
-
Colt Division had the following results for the year just ended: Sales..................................$750,000 Contribution margin ..................300,000 Controllable...
-
Under what circumstances is a shop-floor control system needed?
-
Ellipses Corp is a small business that operates in Herndon, VA. The company is located at10 Period Lane, Herndon, VA 20170. Its federal Employer Identification Number is 77-7777777, and its...
-
Percival Pharmaceuticals manufactures an over-the-counter allergy medication called Breathe. Percival is trying to win market share from Sudafed and Tylenol. The company has developed several...
-
Jason Hand, the new plant manager of Old Tree Manufacturing Plant Number 7, has just reviewed a draft of his year-end financial statements. Hand receives a year-end bonus of 8% of the plants...
-
Draw energy band diagram for a MOS structure in thermal equilibrium with (a). P-type semiconductor. Use4 , 3.5 , 1.0m sc Gq eV q eV E eV = = = and calculate VFB. Estimate the required applied voltage...
-
Classify each function as a power function, root function, polynomial (state its degree), rational function, algebraic function, trigonometric function, exponential function, or logarithmic function....
-
Which one of the following items could be included in the notes to the statements? a. Pending company takeovers b. IFRS or ASPE handbooks c. The competitors' financial statements d. A contract signed...
-
Use the option quote information from Euronext Liffe shown here for Xstrata plc to answer the questions that follow. The equity is currently selling for 11.025. (a) Are the call options in the money?...
-
Ziff Electrics (ZE) is a public utility that provides electricity to the whole Yorkshire region. Recent events at its Mile-High Nuclear Station have been discouraging. Several shareholders have...
-
Consider the following pre-merger information about firm A and firm B: Assume that firm A acquires firm B via an exchange of equity at a price of DKr20 for each share of Bs equity. Both A and B have...
-
The Chocolate Ice Cream Company and the Vanilla Ice Cream Company have agreed to merge and form Fudge Swirl Consolidated. Both companies are exactly alike except that they are located in different...
-
Each month, a company can rent capital for $7,500 per unit and can hire workers for $2,500 each. Currently, the company is using 4 units of capital and 6 workers to produce 10,000 units of output....
-
The correct statement about SEO includes: A. Stock price tend to go up when SEO is issued B. It is one of the important ways for a firm to raise debt C. Like stock buyback, SEO issuing can also be...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
True or False: Inflation decreases the purchasing power of money.
-
Logan is conducting an economic evaluation under inflation using the then-current approach. If the inflation rate is \(j\) and the real time value of money rate is \(d\), which of the following is...
-
True or False: The most commonly used measure of inflation is the relative change in the Consumer Price Index.

Study smarter with the SolutionInn App

