Sulfur tetrafluoride gas is collected at 34.0 C in an evacuated flask with a measured volume...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
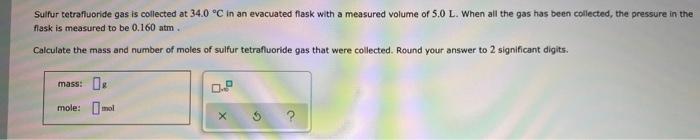
Sulfur tetrafluoride gas is collected at 34.0 °C in an evacuated flask with a measured volume of 5.0 L. When all the gas has been collected, the pressure in the flask is measured to be 0.160 atm. Calculate the mass and number of moles of sulfur tetrafluoride gas that were collected. Round your answer to 2 significant digits. mass: mole: mol 0.P Sulfur tetrafluoride gas is collected at 34.0 °C in an evacuated flask with a measured volume of 5.0 L. When all the gas has been collected, the pressure in the flask is measured to be 0.160 atm. Calculate the mass and number of moles of sulfur tetrafluoride gas that were collected. Round your answer to 2 significant digits. mass: mole: mol 0.P
Expert Answer:
Answer rating: 100% (QA)
2 NoNz 8 2 Na 8 3N gl Sfy sulfur tetrafluoride Molar mass 108 gmol ... View the full answer

Related Book For 

Posted Date:
Students also viewed these general management questions
-
In addition to the data shown in Exhibit 1 and Exhibit 2, what other financial information would you have obtained if you had been responsible for planning the 1991 Leslie Fayaudit? EXHIBIT continued...
-
Quintana Company presents the balance sheet shown in Exhibit 6.30 and the statement of cash flows shown in Exhibit 6.31 for 2013. The firm sold investments, equipment, and land for cash at their net...
-
Look at the balance sheet shown in Exhibit 2.3. Redo that balance sheet in the more traditional format used by Leons. How does your statement differ from the one prepared by the company? Which...
-
Provide the appropriate statute for your answer (format style - IRAC - Issue, Rule, Application and Conclusion.) what are your thought? Problem Scenario- Earnest is married to Janice. Earnest and...
-
Label each of the following as a strong or weak acid or base: a. H3PO4 b. HClO c. HClO4 d. Sr(OH)2
-
What needs to be disclosed according to AASB 12/IFRS 12 for each subsidiary where a non-controlling interest exists?
-
What can be used for a velocity output without the negative aspects of a seismic transducer?
-
You are auditing inventory of HUSKY Corp. as of December 31, 2007.The inventory general ledger balance is $8,124,998.66. HUSKY manufactures lawn and garden tractors, snowmobiles, and supplies....
-
Evaluate the challenges and strategies associated with implementing quality control in complex, global supply chains, considering the impact of multi-tier suppliers, geographical dispersion, and...
-
The cash receipts journal of Silver Plastics follows. Silver s general ledger includes the following selected accounts, along with their account numbers: Indicate whether each posting reference (a)...
-
Global Corporation distributed property with an $664,000 fair market value and a $365,200 adjusted basis to one of its shareholders. The property was subject to a $200,860 mortgage, which the...
-
'Budgeting serves as a benchmark for evaluation.' Explain what this statement means.
-
What is a 'cash buffer' and what is an example of a circumstance where a business could use one?
-
Amy Dixon opened the Dixon Travel Agency in January, and the company entered into the following transactions during January: a On 2 January Amy deposited \(\$ 23000\) in the cash account. b To...
-
If a problem comes to light during the budgeting process, what is the manager likely to do?
-
If you just finished budgeting sales for next year, what information would you need to be able to budget cash collections from sales?
-
Today is the day. Your debate is here and each side will need to make its opening remarks. You are arguing that people who want to stay energized throughout the day should find other ways than using...
-
According to a recent survey, 40% of millennials (those born in the 1980s or 1990s) view themselves more as spenders than savers. The survey also reveals that 75% of millennials view social...
-
Identify the specific circumstances under which auditors are allowed to provide confidential client information to third parties.
-
Once an audit firm has informed client management of important internal control weaknesses, what further responsibility, if any, does the audit firm have regarding those items? For example, does the...
-
Most of Judge Liflands criticism of E&Y focused on the firms audit procedures for CBIs accounts payable. Generally, what is the primary audit objective for accounts payable? Do you believe that E&Ys...
-
With diverse patient populations come language translation issues. Medical interpretation is a challenge facing most health organizations. Medical interpretation and translation services are costly....
-
Visit the Hofstede Centre (https://geerthofstede.com/culture-geerthofstede- gert-jan-hofstede/6d-model-of-nationalculture/) and review the scores by country for the various cultural dimensions that...
-
You have been asked to join the hospitals task force for developing a plan to increase the organizations workforce diversity from its current 20% level to 40% over the next 5 years. How does your...

Study smarter with the SolutionInn App


