You want to construct a portfolio containing equal amounts of U.S. Treasury bills, stock A, and...
Fantastic news! We've Found the answer you've been seeking!
Question:

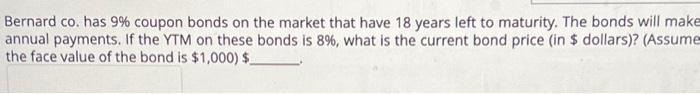
Transcribed Image Text:
You want to construct a portfolio containing equal amounts of U.S. Treasury bills, stock A, and stock B. If the beta of the stock A is 1.23 and the beta of the portfolio is 1.02, what does the beta of stock B have to be? Bernard co. has 9% coupon bonds on the market that have 18 years left to maturity. The bonds will make annual payments. If the YTM on these bonds is 8%, what is the current bond price (in $ dollars)? (Assume the face value of the bond is $1,000) $_ You want to construct a portfolio containing equal amounts of U.S. Treasury bills, stock A, and stock B. If the beta of the stock A is 1.23 and the beta of the portfolio is 1.02, what does the beta of stock B have to be? Bernard co. has 9% coupon bonds on the market that have 18 years left to maturity. The bonds will make annual payments. If the YTM on these bonds is 8%, what is the current bond price (in $ dollars)? (Assume the face value of the bond is $1,000) $_
Expert Answer:
Answer rating: 100% (QA)
i We may begin by using the formula for a portfolios beta which is portfolio wA A wB B where wA wB a... View the full answer

Related Book For 

Posted Date:
Students also viewed these finance questions
-
Here are comparative statement data for Spencer Ltd and Forrester Ltd, two competitors. All data relating to the statement of financial position are as at 30 June, the end of the reporting period for...
-
Emma Lu obtained registration to practice as a naturopath, and spent the month of July 2016 setting up her business E. Lu, Naturopath. Emma prepared a new balance sheet after each transaction which...
-
You have three identical flasks, each containing equal amounts of N2, O2, and He. The volume of the N2 flask is doubled, the O2 flask volume is halved, and the He flask volume is reduced to one-third...
-
Describe the general process used to determine the ULRD using AICPA sampling tables?
-
Give an SQL schema definition for the employee database of Figure. Choose an appropriate domain for each attribute and an appropriate primary key for each relationschema. employee (employee-name,...
-
Calculate the gross profit and operating income for June using absorption costing. Use the following information for Short Exercises Dracut Company reports the following information for June: Net...
-
Kruger Designs hired a consulting firm three months ago to redesign the information system used by the architects. The architects will be able to use state-of-the-art CAD programs to help in...
-
You have risen through the ranks of a coffee company, from the lowly green-apron barista to the coveted black apron, and all the way to CFO. A quick Internet check shows you that your companys beta...
-
Your firm is considering a new three-year project. You know that the unlevered cost of equity for firms with a similar risk as your target is 8%. At the end of the project, all available funds are...
-
Krazy Kars Pty Ltd (Krazy Kars) equips and sells customised off-road vehicles to operators in the tourist industry. The directors are Carlos, Possum and Michele. According to its constitution,...
-
Homework: Assignment # 1 - Network Diagram + Gantt Chart Question 1, Problem 3.8 (book match) > HW Score: 0%, 0 of 26 points Points: 0 of 9 Save Question list K The City of Lethbridge has decided to...
-
The pressure of a gas is given by \(P=\frac{15}{V}\), where \(P\) is in atmosphere and \(V\) is in litres. If the gas expands from 20 to \(60 \mathrm{~L}\) and undergoes an increase in internal...
-
True or False. The rigid body mode is also called the zero mode.
-
What is the importance of latent heat of fusion and vaporization?
-
A piston-cylinder assembly containing a gas undergoes a process in which the temperature of the system rises from \(100^{\circ} \mathrm{C}\) to \(150^{\circ} \mathrm{C}\). The heat transmission per...
-
Establish the ideal gas equation of state from Charles' law, Boyle's law and Avogadro's law. Enumerate the limitation of the ideal gas law.
-
An estimated probability that a Flight leaving Chicago O'Hare airport is 0.45. Some days of the week are prone to longer delays than others. It turns out on Mondays 1400 out of the 2200 flights are...
-
Evaluate the line integral, where C is the given curve. C x 2 dx + y 2 dy, C consists of the arc of the circle x 2 + y 2 = 4 from (2, 0) to (0, 2) followed by the line segment from (0, 2) to (4, 3)
-
List the following compounds in order of increasing acid strength: HBr, H2Se, H2S.
-
Calculate the CH and CCl bond lengths in chloroform, CHCl3, using values for the covalent radii from Table 9.4. How do these values compare with the experimental values: CH, 107 pm; CCl, 177 pm?...
-
Which of the following compounds would you expect to exhibit only London forces? a. Potassium chloride, KCl b. c. Silicon tetrafluoride, SiF4 d. Phosphorus pentachloride, PCl5 (
-
The production budget for cars for May (in units) is a. 6,500 b. 11,000 c. 4,500 d. 10,100
-
The total cost of goods sold for June is a. $103,500 b. $128,800 c. 57,500 d. \($232,300\) Munch Enterprises makes a small toy car that is voice activated. Projected sales for the next four months...
-
What is Munchs expected profit for April? a. $192,500 b. $56,000 c. $66,000 d. $59,000

Study smarter with the SolutionInn App


