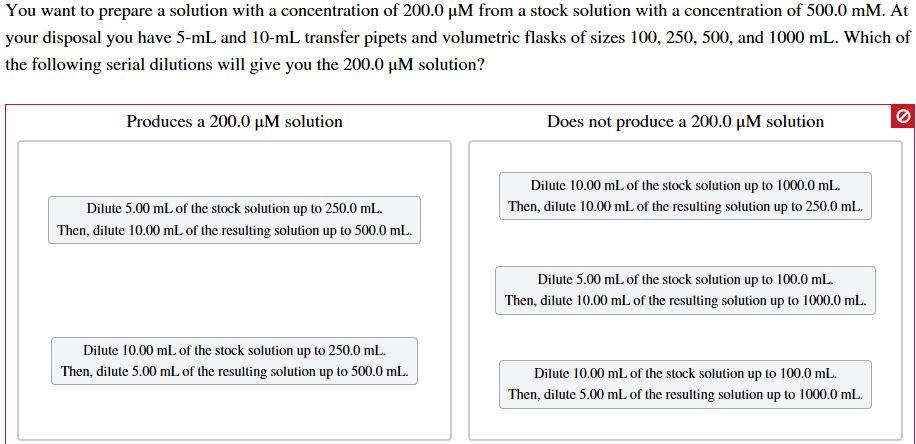
You want to prepare a solution with a concentration of 200.0 M from a stock solution...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
You want to prepare a solution with a concentration of 200.0 µM from a stock solution with a concentration of 500.0 mM. At your disposal you have 5-mL and 10-mL transfer pipets and volumetric flasks of sizes 100, 250, 500, and 1000 mL. Which of the following serial dilutions will give you the 200.0 µM solution? Produces a 200.0 µM solution Does not produce a 200.0 µM solution Dilute 10.00 ml of the stock solution up to 1000.0 mL. Dilute 5.00 mL of the stock solution up to 250.0 mL. Then, dilute 10.00 ml of the resulting solution up to 250.0 ml. Then, dilute 10.00 mL of the resulting solution up to 500.0 mL. Dilute 5.00 ml of the stock solution up to 100.0 mL. Then, dilute 10.00 ml of the resulting solution up to 1000.0 ml. Dilute 10.00 mL of the stock solution up to 250.0 mL. Then, dilute 5.00 mL of the resulting solution up to 500.0 mL. Dilute 10.00 mL of the stock solution up to 100.0 mL. Then, dilute 5.00 mL of the resulting solution up to 1000.0 mL. You want to prepare a solution with a concentration of 200.0 µM from a stock solution with a concentration of 500.0 mM. At your disposal you have 5-mL and 10-mL transfer pipets and volumetric flasks of sizes 100, 250, 500, and 1000 mL. Which of the following serial dilutions will give you the 200.0 µM solution? Produces a 200.0 µM solution Does not produce a 200.0 µM solution Dilute 10.00 ml of the stock solution up to 1000.0 mL. Dilute 5.00 mL of the stock solution up to 250.0 mL. Then, dilute 10.00 ml of the resulting solution up to 250.0 ml. Then, dilute 10.00 mL of the resulting solution up to 500.0 mL. Dilute 5.00 ml of the stock solution up to 100.0 mL. Then, dilute 10.00 ml of the resulting solution up to 1000.0 ml. Dilute 10.00 mL of the stock solution up to 250.0 mL. Then, dilute 5.00 mL of the resulting solution up to 500.0 mL. Dilute 10.00 mL of the stock solution up to 100.0 mL. Then, dilute 5.00 mL of the resulting solution up to 1000.0 mL.
Expert Answer:
Answer rating: 100% (QA)
2 Answer 500 mm Given stock solution Concentration Required Concentration from stock 200 MM ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
You want to prepare a buffer solution that has a pH equal to the pKa of the acid component of the buffer. If you have 100 mL of a 0.10 M solution of the acid HA, what volume and concentration of NaA...
-
You want to prepare 500.0 mL of 1.000 M KNO3 at 20C, but the lab (and water) temperature is 24C at the time of preparation. How many grams of solid KNO3 (density 2.109 g/mL) should be dissolved in a...
-
The BSA standard solution with the concentration of 0.5 mg/mL was used to produce standard curve for protein assay based on Bradford method The test tubes contains BSA solutions was prepared: 0, 20,...
-
ARTICLE: Reference Resolve Asset Management's Yes, You Can Eat Sharpe Ratios. 1- The author spoke to two ways for the foundation to meet its target return of 5%. Identify what the two ways presented...
-
Name and describe three problem-solving tools.
-
Show that the competitive labor market compensates workers for the probability that they will be laid off.
-
A contingency table containing observed values has three rows and four columns. The number of degrees of freedom for the chi-square statistic is 7. In Exercises 68, determine whether the statement is...
-
Donaldson sold plumbing supplies. The St. Paul-Mercury Indemnity Co., as surety for him, executed and delivered a bond to the state of California for the payment of all sales taxes. Donaldson failed...
-
4. A 3 kg copper part, initially at 500 K, is plunged into a tank containing 5 kg of liquid water, initially at 300 K. The copper part and water can be modeled as incompressible with specific heats...
-
Collyer Products Inc. has a Valve Division that manufactures and sells a standard valve as follows: The company has a Pump Division that could use this valve in the manufacture of one of its pumps....
-
Compare and Contrast Leadership Styles and Evaluate the Best Coach Fit After reviewing the links below on coaching leadership styles present the coaches and their style might work best for you if you...
-
The real risk-free rate is 1.56%, inflation is expected to be 3.88% this year, and the maturity risk premium is zero. What is the equilibrium rate of return on a 1-year Treasury security? (Express...
-
The Coffee Table Company Ltd was established on 31st December 2005 to manufacture coffee tables for sale to the furniture retail trade. The owners invested K100 million as share capital. A long term...
-
List and discuss the handwriting on the wall. How does Who Moved my Cheese help you deal with obstacles and change?
-
An investment group of 15 college students at Duke University decided to contribute $ 255 each into a regular savings account. Determine how much would be in the account after4 years at 1.23%...
-
In what ways would transactional and laissez-faire leadership styles be ineffective in encouraging organizational commitment? How might leadership style need to change with an organization's...
-
4. By hand, compute the ALU decomposition of the following 4 x 4 matrix A 1-2-2-3] 3 -9 -95 -1 2 4 7 -3 -6 24 3 Be sure to include your working for each step of the procedure, including the forms of...
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
What is the pH of a buffer solution that is 0.15 M chloroacetic acid and 0.10 M sodium chloroacetate? Ka = 1.3 10-3.
-
A sample of sodium thiosulfate, Na2S2O3, contains sulfur-35. Determine the mass of 35S in the sample from the decay rate, which was determined to be 7.7 1011 disintegrations per second. The...
-
If adenine, thymine, guanine, and cytosine were each analyzed separately in a sample of DNA, what molar ratios of A:T and G : C would you expect to find?
-
What is automatic control?
-
A block diagram is represented by (a) circles (b) triangles (c) rectangles (d) parallelograms
-
What is a transducer? Explain the principle of operation of a transducer.

Study smarter with the SolutionInn App


