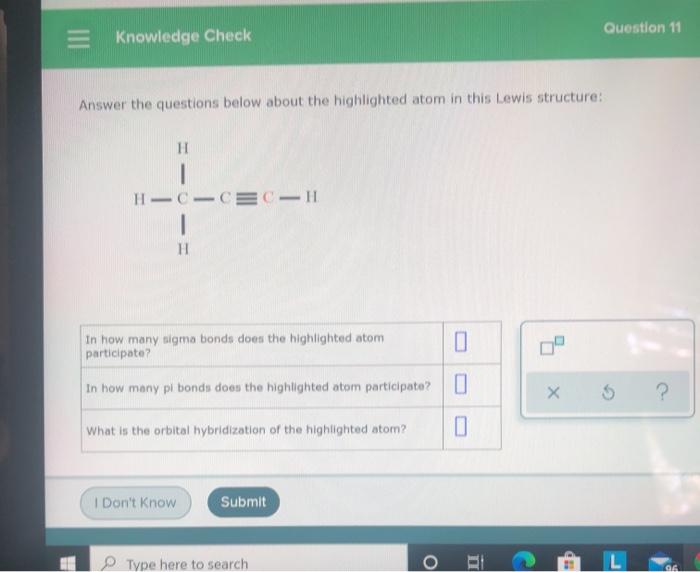
= Knowledge Check Answer the questions below about the highlighted atom in this Lewis structure: H...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
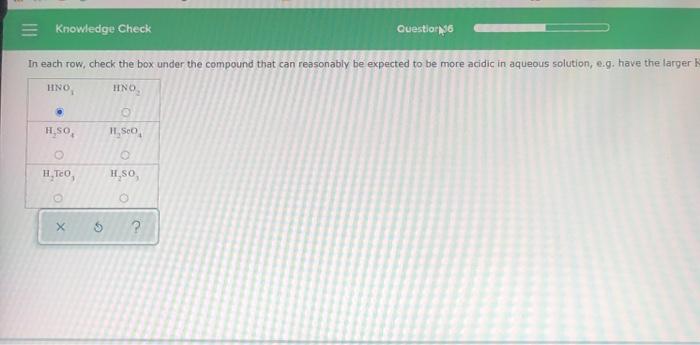
= Knowledge Check Answer the questions below about the highlighted atom in this Lewis structure: H HICICCIH H In how many sigma bonds does the highlighted atom participate? In how many pi bonds does the highlighted atom participate? What is the orbital hybridization of the highlighted atom? I Don't Know Submit Type here to search O 0 0 n Question 11 XS ? L 96 Knowledge Check In each row, check the box under the compound that can reasonably be expected to be more acidic in aqueous solution, e.g. have the larger K HNO, HINO H.SO, H₂TeO, O X O H₂SO O H.SO, O Question 36 ? = Knowledge Check Answer the questions below about the highlighted atom in this Lewis structure: H HICICCIH H In how many sigma bonds does the highlighted atom participate? In how many pi bonds does the highlighted atom participate? What is the orbital hybridization of the highlighted atom? I Don't Know Submit Type here to search O 0 0 n Question 11 XS ? L 96 Knowledge Check In each row, check the box under the compound that can reasonably be expected to be more acidic in aqueous solution, e.g. have the larger K HNO, HINO H.SO, H₂TeO, O X O H₂SO O H.SO, O Question 36 ?
Expert Answer:
Answer rating: 100% (QA)
1 highlighted C forms 2 sigma bonds 1 sigma bonds with each carbon atom Answ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
You want to prepare 500.0 mL of 1.000 M KNO3 at 20C, but the lab (and water) temperature is 24C at the time of preparation. How many grams of solid KNO3 (density 2.109 g/mL) should be dissolved in a...
-
You want to prepare a buffer solution that has a pH equal to the pKa of the acid component of the buffer. If you have 100 mL of a 0.10 M solution of the acid HA, what volume and concentration of NaA...
-
Water is to be boiled at sea level in a 30-cm-diameter stainless steel pan placed on top of a 3kW electric burner. If 60 percent of the heat generated by the burner is transferred to the water during...
-
The Regal Cycle Company manufactures three types of bicycles-a dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Sales Variable manufacturing and...
-
You have been retained by a major entertainment company to evaluate the purchase of land near one of its new amusement parks in Australia. The land will not be developed immediately, but it will be...
-
Efficiency of closed cycle gas turbine as compared to open cycle gas turbine is: (a) More (b) Less (c) Same (d) None of the above
-
Why would you also want to check social media sites for information about your own client or witness?
-
Butler Manufacturing Corporation raised capital for a plant expansion by borrowing from a bank and making a stock offering. Butler engaged Weaver, CPA, to audit its December 2014 financial...
-
Built-Tight is preparing its master budget. Budgeted sales and cash payments follow: Budgeted sales. July $ 57,500 August $ 73,500 Budgeted cash payments for Direct materials Direct labor Overhead...
-
A textile company produces shirts and pants. Each shirt requires two square yards of cloth, and each pair of pants requires three square yards of cloth. During the next two months the following...
-
1. Cultural differences are often expressed in the "generation gap". Choose one each from the list of symbols mentioned that you and your parents share and believe in (e.g. religion, education, and...
-
Who is the major player in installing databases? What are the responsibilities?
-
What are some potential problems of using parallel conversion as a conversion strategy?
-
Sketch an isentropic and an actual turbine process on a Ts diagram when the inlet flow is a superheated vapor and the outflow is a saturated mixture. Include the vapor dome and the pressure contours...
-
What are some potential problems of using abrupt cut-over as a conversion strategy?
-
Sketch a throttling process of an ideal gas on a Ts diagram. Include the pressure contours that pass through the initial and final states.
-
1.2 A Grade 2 class is struggling with the following flow diagram. A B C D E 50 52 +9 54 67 68 They provided their answers as: A: 59; B: 61; C: 63; D: 76; E: 77. Identify the mistake that the Grade 2...
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
Distinguish Lewis acids and bases from Bronsted-Lowry acids and bases. Give an example of each.
-
Identify the Brnsted-Lowry acids among the reactants in the following reactions: a. b. KCN + HI = HCN + KI
-
What color would you expect to observe for a solution of Fe(ferrozine) 4 3 - which has a visible absorbance maximum at 562 nm?
-
United Technologies Corporation (UTC), based in Hartford, Connecticut, owns a wide variety of companies that operate in different businesses and industries. Some of the companies in UTCs portfolio...
-
How does the auditors professional duty of confidence affect disclosures to third parties?
-
When can auditors disclose unlawful acts to the police?

Study smarter with the SolutionInn App


