ZnCl Ph Ni(BF4) EtO Ph A Ph3C. Suggest a reasonable mechanism for the following reaction Ph...
Fantastic news! We've Found the answer you've been seeking!
Question:
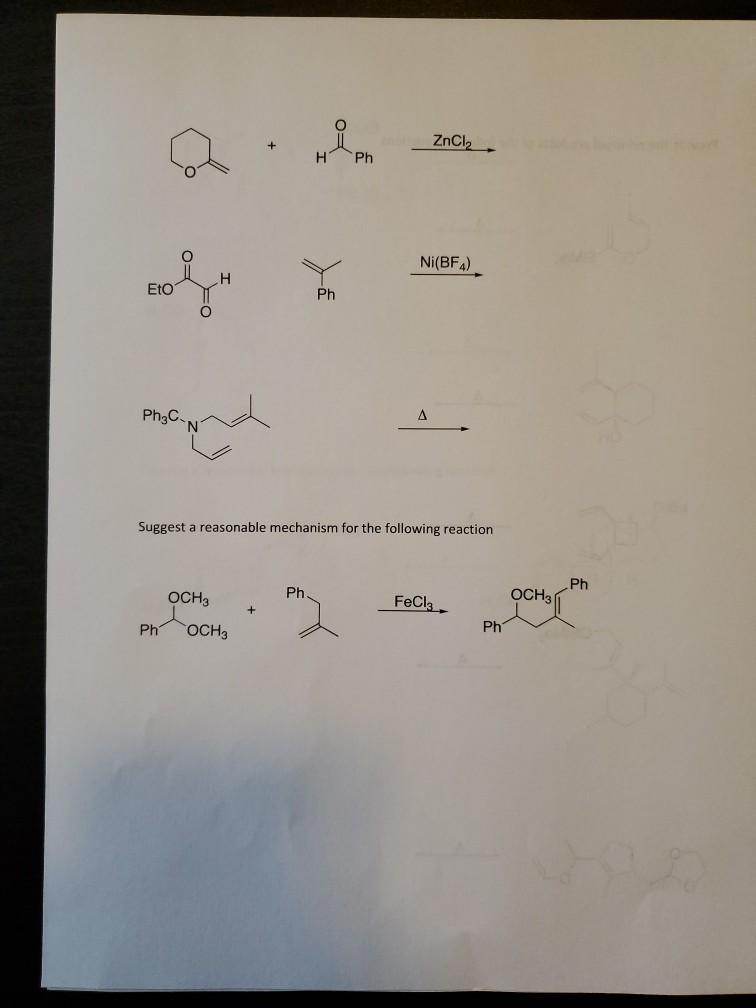
Transcribed Image Text:
ZnCl Ph Ni(BF4) EtO Ph A Ph3C. Suggest a reasonable mechanism for the following reaction Ph OCH3 Ph. OCH3 FeCl, Ph Ph OCH3 ZnCl Ph Ni(BF4) EtO Ph A Ph3C. Suggest a reasonable mechanism for the following reaction Ph OCH3 Ph. OCH3 FeCl, Ph Ph OCH3
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Suggest a reasonable mechanism for each of the following reactions: (a) (b) (c) (d) I. excess NaNH, NH C,H,Br CH(COOCH,CH)OoCH,CH,) l excess NH 2.,0 CH2CH2CH2CH2COCH2CH3 NaNH Cl COOCH CH CH3 CH3...
-
Suggest a reasonable mechanism for each of the following reactions:
-
The equilibrium constant for the following reaction is 1.0 Ã 10-3: Cr'. (aq) + H2EDTA2-(aq)--CrEDT A-(aq) + 2H' (aq) CH2-CO2 02GH CH EDTA N-CH,--CH2- O2C-CH2 CHy-CO2-...
-
One end of a light, elastic string, of natural length 1.2m and modulus of elasticity 32N, is attached to a fixed point, B. A particle, P, of mass 1.5 kg, is then attached to the other end of the...
-
Prepare journal entries to record the following transactions by Ericton Industries: 2014 Jan. 20 Accepted a $ 170,000, 90-day, 9% note dated this day in granting a time extension on the past due...
-
You decide to set up a college fund for your 10-year-old child and plan to make annual deposits into the account each year on your child's birthday. Because "other things" consistently use more of...
-
A gas engine working on Otto cycle has a cylinder of diameter \(220 \mathrm{~mm}\) and stroke \(300 \mathrm{~mm}\). The clearance volume is \(1600 \mathrm{cc}\). Find the air-standard efficiency....
-
Using the mutual fund quotes in Exhibit 13.4, and assuming that you can buy these funds at their quoted NAVs, how much would you have to pay to buy each of the following funds? Artio Select...
-
5. [10 points] Consider the following graph: 2 5 3 6 a) Starting at vertex 1 and resolving ties by vertex d number, traverse the graph by depth- first search and construct the corresponding...
-
How can I find the exact p-value for each coefficient (p>t )column if the number of observations = 52, df numerator is 3, df denominator is 48, t static is there in the third column. I need to know...
-
Suppose a 10-year project requires the following initial investment costs. Building: -Lasts for 10 years - Initial cost of $60,000 Equipment - Last for 8 years - Initial cost $15,000 Vehicles: - Last...
-
Why do some people dread public speaking or just want to avoid it? Identify some potential benefits of public speaking in academic, professional, personal, and civic contexts that might make people...
-
Tara and Todd are married. Tara is a lawyer who operates her practice as a sole proprietorship. Todd works for a local architecture firm. Their taxable income before the QBI deduction is $404,200,...
-
The following data refer to Huron Corporation for the year 20x2. Sales revenue Raw-material inventory, 12/31/x1 Purchases of raw material in 20x2 Raw-material inventory, 12/31/x2 Direct labor cost...
-
A mineral processing plant will soon undergo a shutdown for a major annual maintenance. The shutdown will last a total of eight days, and will require a significant number of contract workers in each...
-
Waterway Company manufactures two products, Mini A and Maxi B. Waterway's overhead costs consist of setting up machines- $870000; machining- $2040000; and inspecting- $610000. Information on the two...
-
If x(x+1) greater than 0, does it follow that x is positive? If no give an example
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
(a) What compound of molecular formula C6H10 gives 2,3-dimethylbutane on catalytic hydrogenation over platinum? (b) What two compounds of molecular formula C11H20 give 2,2,6,6-tetramethylheptane on...
-
Write the structure of the major organic product isolated from the reaction of 1-hexyne with (a) Hydrogen (2 mol), platinum (b) Hydrogen (1 mol), Lindlar palladium (c) Lithium in liquid ammonia (d)...
-
Most of the NMR spectra in this text were recorded on a spectrometer having a field strength of 4.7 T (200 MHz for 1H). The first generation of widely used NMR spectrometers were 60-MHz instruments....
-
The following is the distribution of the hourly number of trucks arriving at a company's warehouse: Find the mean of this distribution, and using it (rounded to one decimal place) as the parameter...
-
With reference to Exercise 10.42, verify that the mean of the observed distribution is 1.6 , corresponding to \(40 \%\) of the cars requiring repairs. Then look up the probabilities for \(n=5\) and...
-
Among 100 purification filters used in an experiment, 46 had a service life of less than 20 hours, 19 had a service life of 20 or more but less than 40 hours, 17 had a service life of 40 or more but...

Study smarter with the SolutionInn App



