As shown in Fig. 16-2, two flasks are connected by an initially closed stopcock. One flask contains
Question:
As shown in Fig. 16-2, two flasks are connected by an initially closed stopcock. One flask contains krypton gas at 500 mmHg, while the other contains helium at 950 mmHg. The stopcock is now opened so that the gases mix. What is the final pressure in the system? Assume constant temperature.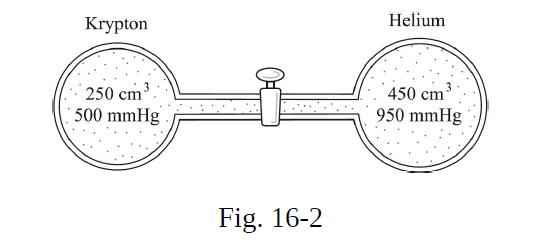
Transcribed Image Text:
Krypton 250 cm 500 mmHg 여 Fig. 16-2 Helium 450 cm 950 mmHg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
According to Boyles Law the pressure of a gas is inversely proportional to its volume at constant ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
In what ways is this issue similar to and different from controversies about using illegal aliens to perform low-wage jobs inside the U.S.?
-
As shown in fig 1.7, a manometer is attached to a tank of gas in which the pressure is 104.0 kPa. The manometer liquid is mercury, with a density of 13.59 g/cm3. If g = 9.81 m/s2 and the atmospheric...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Find a vector parametrization of the intersection of the surfaces x 2 + y 4 + 2z 3 = 6 and x = y 2 in R 3 .
-
Discuss why many information technology professionals may overlook project cost management and how this might affect completing projects within budget.
-
In 2019, Hunter and Monda (both under age 50) had compensation income of $1,000,000 and $200,000, respectively. Adjusted gross income on their joint return was $1,200,000, and neither taxpayer was a...
-
Although the customer loyalty project at Petrie Electronics had gone slowly at first, the past few weeks had been fast-paced and busy, Jim Watanabe, the project manager, thought to himself. He had...
-
Hearty Soup Co. uses a process cost system to record the costs of processing soup, which requires the cooking and filling processes. Materials are entered from the cooking process at the beginning of...
-
How does a Cloud Data Management Platform differ from a Database Management System?
-
A 500-mL sealed flask contains nitrogen at a pressure of 76.00 cmHg. A tiny glass tube lies at the bottom of the flask. Its volume is 0.50 mL and it contains hydrogen gas at a pressure of 4.5 atm....
-
One method for estimating the temperature at the center of the Sun is based on the Ideal Gas Law. If the center is assumed to consist of gases whose average M is 0.70 kg/kmol, and if the density and...
-
Write the atomic symbol for the element whose monatomic ion has a 2 + charge, has 14 more neutrons than electrons, and has a mass number of 88.
-
Gypsy has used the same insurance company for her car insurance for 10 years. She always gets coverage for $500,000 and pays her premium on time. Her insurance policy was just renewed. She kept on...
-
Explain why it's important to identify and classify all our stakeholders? Provide an example of what could happen if you fail to identify a stakeholder . Explain what information the stakeholder...
-
The following selected information (in thousands) is available for Cullumber Inc.: Total assets Total liabilities Interest expense Income tax expense Profit for the year Cash provided by operating...
-
For CAPSIM Simulation, why was our plant utilization so low? Our days of working capital in round 4, why have days of working capital decreased? Please explain more analysis. Internal Business...
-
You learned about the different marketing reasons why new products fail. Identify three (3) reasons why Earth's Own Oat Milk has been a success and explain your answer.
-
Wilmington Pharmacy, part of a large chain of pharmacies, fills a variety of prescriptions for customers. The complexity of prescriptions filled by Wilmington varies widely; pharmacists can spend...
-
A 6-lb shell moving with a velocity ?? v0k explodes at point D into three fragments which hit the vertical wall at the points indicated. Fragments A, B, and C hit the wall 0.010 s, 0.018 s, and 0.012...
-
An object is located at a distance s 0 from a spherical mirror of radius R.Show that the resulting image will be magnified by an amount R MT = 2s, + R
-
Design a little dentists mirror to be fixed at the end of a shaft for use in the mouth of some happy soul. The requirements are (1) that the image be erect as seen by the dentist and (2) that when...
-
An LED 0.60 cm tall is on the central axis 30.0 cm in front of a convex spherical mirror. If the radius of curvature of the mirror is 12.0 cm determine the location of the image, describe it, and...
-
You (Donna) are a staff auditor in your second year with a public accounting firm. This week you have been assigned to work on the BBB audit, which is already nearing completion. The staff auditor...
-
To pay an entered bill from a supplier, you would: (1 Point) Write a cheque Use the Pay Bills window Use the General Journal Create a credit memo 17.It contains a file for each customer with whom the...
-
Identify a significant current issue faced by internal auditing (one issue only) Some general ideas for your consideration are as follows: -Challenges to the business resulting from COVID-19...

Study smarter with the SolutionInn App


