Acetone can be recovered from air by absorption in water. The conditions for the streams entering and
Question:
Acetone can be recovered from air by absorption in water. The conditions for the streams entering and leaving are listed below. If the absorber operates adiabatically, obtain the temperature of the exiting liquid phase using a simulation program.
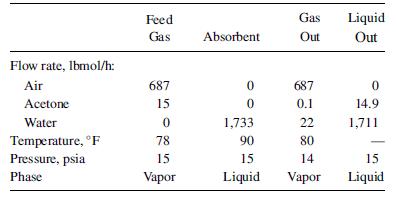
Transcribed Image Text:
Flow rate, lbmol/h: Air Acetone Water Temperature, F Pressure, psia Phase Feed Gas 687 15 0 78 15 Vapor Absorbent 0 0 1,733 90 15 Liquid Gas Liquid Out Out 687 0.1 22 80 14 Vapor 0 14.9 1,711 15 Liquid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
The temperature of the exiting liquid phase can be obtained by running ...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
Acetone, a valuable solvent, can be recovered from air by absorption in water or by adsorption on activated carbon. If absorption is used, the conditions for the streams entering and leaving are as...
-
Water is used to remove CO2 from air by absorption in a column packed with Pall rings. At a certain region of the column where the partial pressure of CO2 at the interface is 150 psia and the...
-
A pump operates adiabatically with liquid water entering at T1 and P1 with a mass flow rate m. The discharge pressure is P2 and the pump efficiency is . For one of the following sets of operating...
-
Adjusting and Closing Presented on the next page is the trial balance of the Crestwood Golf Club, Inc. as of December 31. The books are closed annually on December 31. Instructions (a) Enter the...
-
Egeland et al. (1995) use capturerecapture to estimate the total number of fetal alcohol syndrome cases among Alaska natives born between 1982 and 1989. Two sources of cases were used: thirteen cases...
-
Which of the following statements is incorrect? A. In practice, the calculation of economic profit is determined based on GAAP. B. The market value added is the difference between the market value of...
-
What three factors change air density?
-
The Fraser Paper Company produces large rolls of white paper weighing 1,000 kilograms for wholesalers for $1,500 each. The wholesalers then cut the paper into standard-sized sheets and package it in...
-
The total payroll of Crane Company for October, 2025 was $788000, of whic $147000 represented amounts paid in excess of $128,400 to certain employees for FICA. $490000 represented amounts paid to...
-
For jet aircraft flying out of Houston on a summer afternoon (T = 95 F, 70% relative humidity) and Palm Springs on a winter morning (T = 40 F, 55% relative humidity), determine for both locations: a)...
-
What is the critical solution temperature?
-
What is a minimum-boiling azeotrope? What is a maximum- boiling azeotrope? Which type is by far the most common?
-
Discuss how transhumance herders graze their livestock.
-
Refer to the previous exercise. Determine your profit if at the expiration date the settle price of the contract is 135 points. Calculate the gain or loss in each position. Previous exercise...
-
Graph the solution of each system given in Problems 5-18. \(\left\{\begin{array}{l}3 x-2 y \geq 6 \\ 2 x+3 y \leq 6\end{array} ight.\)
-
Sketch the graph of each equation in Problems 3-30. \(y=\frac{1}{10} x^{2}\)
-
Graph the first-degree inequalities in two unknowns in Problems 13-48. \(y>5 x-3\)
-
Graph the lines, curves, or half-planes in Problems 1-12. \(2 x+3 y=15\)
-
By how much does the ball in Example B clear a 10 ft fence that is 365 ft away if the wind is blowing directly from the fence toward Carolina at 8 mi/h?
-
In what ways does a well-designed enterprise search software vary from popular search engines (e.g., Bing, DuckDuckGo, and Google)?
-
Myosin II has a duty ratio of 10 percent, and its step sizeis 8 nm. In contrast, myosin V has a much higher duty ratio (about 70 percent) and takes 36-nm steps as it walks down an actin filament....
-
Contraction of both skeletal and smooth muscle is triggered by an increase in cytosolic Ca 2+ . Compare the mechanisms by which each type of muscle converts a rise in Ca 2+ into contraction.
-
Phosphorylation of myosin light-chain kinase (MLCK) hy protein kinase A (PKA) inhibits MLCK activation by Ca 2+ calmodulin. Drugs such as albuterol bind to -adrenergic receptor, which cause a rise in...
-
1. Write a Java program that will prompt the user for a number and print out a square with those dimensions. For example, if the user enters 5, return the following: * * * ** ** * * * * * * * * * * *...
-
2. Vector multiplication or dot product is performed by multiplying corresponding elements and summing the products. It can be represented mathematically as oa; *bi where a and b are vectors of...
-
The following data was collected from an experiment testing the hypothesis: The density of cream whipped for 5 minutes increases at higher altitudes. What would be an acceptable interpretation of...

Study smarter with the SolutionInn App


