Ten thousand kg/h of a 10 wt% liquid solution of naphthalene in benzene is cooled from 30
Question:
Ten thousand kg/h of a 10 wt% liquid solution of naphthalene in benzene is cooled from 30ο C to 0οC. Determine the amount of crystals and composition and flow rate of the mother liquor. Are the crystals benzene or naphthalene? Use Figure 4.22.
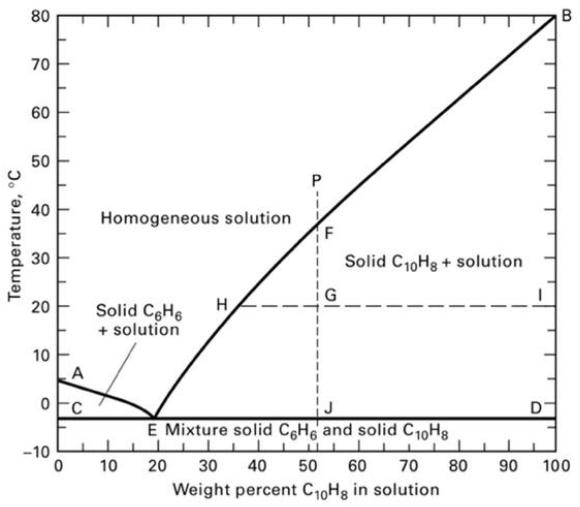
Transcribed Image Text:
Temperature, °C 80 70 60 50 40 30 20 10 A oc -10 0 Homogeneous solution Solid C6H6 + solution 10 H F Solid C₁0Hg + solution E Mixture solid C6H6 and solid C10H8 L 20 30 40 50 60 70 80 90 Weight percent C₁0H8 in solution D B 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Answer The amount of crystals would be approximately 1429 kg...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Separation Process Principles Chemical And Biochemical Principles
ISBN: 9780470481837
3rd Edition
Authors: By J. D. Seader, Ernest J. Henley, D. Keith Roper
Question Posted:
Students also viewed these Life Sciences questions
-
solution of Na2SO4. Determine the composition and flow rate of the product if the flow rate of Na2SO4 is 1275 kg/hr, and the ratio of the flow rate of the H2O to the product solution is 0.83.
-
Use figure 22 to find all angles between 0 and 2Ï satisfying the given condition. 9. cos θ = 1 11. tan θ = -1 13. sin x = 23/2 3 12 2 3r 4 2 2 612'2 0 (0,0) 11 7A...
-
Ten kg of a liquid occupies 8000 cm3. Its density, specific volume, and specific gravity are (p, v, SG): (A) (1250 kg/m3, 0.0008 m3/kg, 1.25) (B) (0.0008 kg/m3, 1250 m3/kg, 0.8) (C) (1250 kg/m3,...
-
15. Show that the frequency of revolution of the electron in the Bohr model hydrogen atom is given by v = 2|E|/hn where E is the total energy of the electron. 16. Show that for all Bohr orbits the...
-
Show that (6.28) and (6.29) are equivalent when an SRS of psus is selected as in Chapter 5. Are they equal if psus are selected with unequal probabilities? Discuss.
-
Calculate the mean free time \(\tau\) between the collisions for the conduction electron in copper. \(\left[n=8.49 \times 10^{28} \mathrm{~m}^{-3} ight.\), \(\left.ho_{\mathrm{Cu}}=1.68 \times...
-
Explain the working of an absorption type dynamometer.
-
A process has the transfer function, where K has a nominal-value of K = 1. PID controller settings are to be calculated using the Direct Synthesis approach with ?c = 5 min. Suppose that these...
-
Discuss the principles of fluid instabilities and flow instability mechanisms, including Rayleigh-Taylor instability, Kelvin-Helmholtz instability, and Richtmyer- Meshkov instability, and their role...
-
Company A is a global company based in the United States that operates in the financial industry. Company A serves its customers with financial products, such as checking accounts, bank cards, and...
-
At 20 C, for 1,000 kg of a mixture of 50 wt% Na 2 SO 4 .10H 2 O and 50 wt% Na 2 SO 4 crystals, how many kg of water must be added to completely dissolve the crystals if the temperature is kept at...
-
Figure 7.47 depicts a propylene/propane distillation. Two sievetray columns in series are used because a 270-tray column poses structural problems. Determine column diameters, tray efficiency using...
-
What is a multi-domestic business strategy? What are its advantages?
-
The Church of God at Southaven entered into an agreement with a general contractor called National Church Services (NCS) for the construction of a sanctuary for about \($1.1\) million. NCS hired...
-
The coil in a generator has 100 windings and a crosssectional area of \(0.0100 \mathrm{~m}^{2}\). (a) If the coil turns at a constant rotational speed and the magnetic field in the generator is that...
-
Please calculate the skewness for the rates of return for JNJ and S&P 500 during the period 19892009. Refer to Table 2.4, in which data on rates of return for JNJ, Merck, and S&P 500 during the...
-
Define a vertical restraint on trade, and contrast this definition with that of a horizontal restraint on trade. Which type of restraint is considered to be a greater threat to competition under the...
-
If you wanted to expand your business to include international customers and vendors, what factors would influence your decision?
-
Give the coordinates of the y-intercept of each graph. a. b. c. d. -intercept: 103 r-intereept: 108 -50 1sp -intereept:-100 -inlercept:-90
-
A survey of 70 college freshmen asked whether students planned to take biology, chemistry, or physics during their first year. Use the diagram to answer each question. How many of the surveyed...
-
Which two age groups experienced the highest increase in the number of influenza- and pneumoniarelated deaths between 1917 and 1918? a. How many times greater is the 1918 total than the 1917 total...
-
Although both of these strategies are examples of artificial selection of crop plants, describe two differences between these approaches.
-
Scientists were puzzled why the 1918 flu resulted in increased deaths in specific age groups. What was so unexpected?
-
Having a bit of trouble completing part of the code for my guessing game in java. Basically, I need to add the part of the code that will allow the user to choose how many games they wish to play....
-
Find all the "daffodil numbers" between 100 and 999 and output them. "Daffodil number" refers to a three-digit number, and the cube of each digit is exactly equal to the number itself. For example,...
-
Complete the program so that each cell of array sum contains the sum of the corresponding cells of valA and valB: class Exercise3 { public static void main(String[] args) { int[] valA = {13, -22, 82,...

Study smarter with the SolutionInn App


