Assume that bcc iron at 1800 K has a fraction of vacant Fe sites of 0.0001, an
Question:
Assume that bcc iron at 1800 K has a fraction of vacant Fe sites of 0.0001, an Fe atom vibration frequency
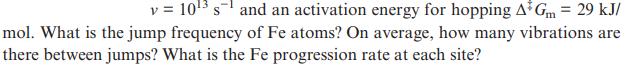
Transcribed Image Text:
v = 10 s and an activation energy for hopping A Gm = 29 kJ/ mol. What is the jump frequency of Fe atoms? On average, how many vibrations are there between jumps? What is the Fe progression rate at each site?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Equation 39 Fe atom has N 8 nearest neighbors The jump frequenc...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Calculate the self-diffusion coefficient of Fe in Problem 3.22, given the unit-cell parameter a = 2.87 . Problem 3.22 Assume that bcc iron at 1800 K has a fraction of vacant Fe sites of 0.0001, an Fe...
-
A BCC iron structure is to be manufactured that will allow no more than 50 g of hydrogen to be lost per year through each square centimeter of the iron at 400 C. If the concentration of hydrogen at...
-
Iron and vanadium both have the BCC crystal structure and V forms a substitutional solid solution in Fe for concentrations up to approximately 20 wt% V at room temperature. Determine the...
-
The compound bow in the previous problem requires 152 J of work to draw the string back to x = 0.540 m, at which point the archer need only exert 250 N of force to hold the string in place. (a)...
-
Determine the polar moment of inertia and the radius of gyration of the isosceles triangle shown with respect to point O.
-
A stock has an expected return of 12 percent and a beta of 1.4, and the expected return on the market is 10 percent. What must the risk-free rate be?
-
A piece of cloth is discovered in a burial pit in the southwestern United States. A tiny sample of the cloth is burned to CO 2 , and the 14 C/ 12 C ratio is 0.250 times the ratio in todays...
-
Apachi Company ended its fiscal year on July 31, 2010. The company??s adjusted trial balance as of the end of its fiscal year is as shown at the top of page 182. Instructions(a) Prepare the closing...
-
A researcher collected self-report data on life satisfaction, social support, and depression. She found the following correlations: (1) life satisfaction and depression, (8) = -.77, p < .01; (2)...
-
Given the diffusivity DC = 10 10 m 2 /s for interstitial carbon in bcc iron at 800 C of a= = 103 S-) 2.87 , estimate the activation energy A Gm for hopping of the C atoms (v= via interstitial sites.
-
Calculate the jump frequency of interstitial carbon in bcc iron at 800 C, assuming a vibration frequency v = 10 s and an activation energy for hopping A*Gm 62 kJ/mol. On average, every x-th carbon...
-
Ringgold NV has outstanding at December 31, 2020, 50,000 shares of 20 par value, cumulative, 6% preference shares and 200,000 shares of 5 par value ordinary share. All shares were outstanding the...
-
When determining depreciation, Expedition followed company policy and relied on Mel Bauer's consulting report for determining the asset's lifetime and salvage value. For the 20X7 calendar year,...
-
Describe the basic tax and nontax characteristics of the primary types of business formations.
-
1. i. Show that given a linear network with a transfer function H(f), the output response is given by y(t) = v(t) *h(t) where v(t) is the input to the network and h(t) its impulse response. [4 marks]...
-
Explain the logic behind the IRS rule why there is no recognized gain under section 3 5 4 ( a ) 1?
-
Assume the spot exchange rate is AUD 0.7030. The expected inflation rate is 1 percent in Australia and 1.3 percent in the U.S. What is the expected exchange rate one year from now if relative...
-
DCL Industries purchased a supply of mechanical components from E Corporation on November 1, 2011. In payment for the $48,000 purchase, DCL issued a 1-year installment note to be paid in equal...
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
Compare the following three isomeric dienes: (a) Which compound will liberate the least heat upon hydrogenation with 2 mol of hydrogen gas? Why? (b) Which compound will liberate the most heat upon...
-
Identify the most stable compound:
-
Draw an energy diagram showing the relative energy levels of the MOs for 1,3,5,7- octatetraene and identify the HOMO and LUMO for both the ground state and the excited state.
-
Consider the seven data samples shown in the following table, where x is the attribute and y; E {-1,1} is the class label of interest i 1 2 3 4 5 6 7 xi -3.0 -1.5 -0.2 0 0.5 2 2.5 yi 1 1 -1 -1 -1 1 1...
-
What is the best O-notation for the following code fragment: for (i=0; i
-
2. Convert the following C instructions to LEGV8 assembly language assume that the variables are already loaded into registers. Specify the registers you will use. [3] a = (h+90) / 1024

Study smarter with the SolutionInn App


