CeMgAl 11 O 19 :Tb 3+ is a green phosphor used in tricolor fluorescent lights. Figure 7.25
Question:
CeMgAl11O19:Tb3+ is a green phosphor used in tricolor fluorescent lights.
Figure 7.25
Transcribed Image Text:
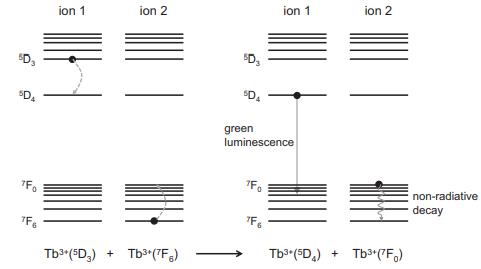
Ce+ ions in the host absorb photons from the Hg-plasma discharge with an excitation max- imum near 270 nm and emit via a [Xe]4f05d [Xe]4f transition with a Stokes shift of 0.9 eV. The Tb+ dopant emits via f-to-f transitions from a D, excited state to various F, levels (see Figure 7.25), with a maximum near 540 nm. (a) At what wavelength will the emission maximum of the Ce+ ion fall? (b) Which lanthanoid ion is responsible for the green light? Why is the other lanthanoid ion needed? (c) What mechanism is responsible for energy transfer between Ce+ and Tb*?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The absorption maximum of 270 nm corresponds to an e...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Create a conclusion to following work. Hotels form an uninterrupted narrative of a story, filling guests with emotions that define their stay. They have ceased to be considered one-dimensional places...
-
A university campus has 200 classrooms and 400 faculty offices. The classrooms are equipped with 12 fluorescent tubes, each consuming 110 W, including the electricity used by the ballasts. The...
-
A university campus has 200 classrooms and 400 faculty offices. The classrooms are equipped with 12 fluorescent tubes, each consuming 110 W, including the electricity used by the ballasts. The...
-
Robert is the owner of an automobile manufacturing company. He calls for a board meeting and tells his directors that he wants to build a car that lets the users experience power and exhilaration. He...
-
A 10-V battery supplies a constant current of 0.5amp to a resistance for 30 min (a) Determine the resistance, in ohms, (b) For the battery, determine the amount of energy transfer by work, in kJ.
-
Determine the tax liability, marginal tax rate, and average tax rate (rounded to two decimal places) in each of the following cases. Use the Form 1040EZ Tax Tables to determine tax liability. a....
-
What is the internal rate of return of the following cash flow diagram? a. 20 percent b. 18.2 percent c. 17.5 percent d. 15 percent $30 $31 0 1 2 3 $30 $15
-
Adria Lopez, owner of Success Systems, decides to prepare a statement of cash flows for her business. (Although the serial problem allowed for various ownership changes in earlier chapters, we will...
-
Consider two oppositely charged atoms with charges of +1 and -1 units, respectively. The two atoms interact with each other through electrostatic (Coulombic) and van der Waals forces, and reach an...
-
When Ce 3+ ions substitute for La 3+ in a LaCl 3 host, the lowest-energy peak in the photoluminescence excitation spectrum falls at 281 nm, whereas a similar substitution in the double-perovskite...
-
The phosphor BaMgAl 10 O 17 :Eu 2+ ,Mn 2+ , whose excitation and emission spectra are shown below, is of interest as a combined blue and green phosphor in plasma-display panels. The Eu 2+ ions absorb...
-
What was the mean score for self-care for each group? Chung and Hwang (2008) examined the difference between an experimental and a control group of patients with leukemia. The experimental group...
-
2. Write a query to display the checkout number, check out date, and due date for all books that has ever been checked out, sorted by checkout number. (See the figure below for first part of the...
-
The purpose of the Replacement Cost Optional Coverage in the Building and Personal Property Coverage Form is to Adjust cost to account for inflation Change valuation basis for loss settlement...
-
In preparing the auditor's report, the report shall contain either an expression of opinion regarding the financial statements or an assertion that an opinion cannot be expressed. identify...
-
Carl delivers papers for spending money. He gets $10 for each day that he works in addition to $0.50 for each paper that he delivers. If Carl works on Monday and gets paid $25, how many papers did he...
-
Jayanti serves multiple roles in SETU as a bookkeeper, management accountant, treasurer, and also an actor. Based on IMA Statement standard of definition of conflict ofinterest, can Jayanti be...
-
Presented below are a number of balance sheet items for Letterman, Inc., for the current year, 2008. Instructions Prepare a classified balance sheet in good form. Common stock authorized was 400,000...
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
A sample containing 2.50 mol of an ideal gas at 325 K is expanded from an initial volume of 10.5 L to a final volume of 60.0 L. Calculate G and A for this process for a. An isothermal reversible path...
-
Why does the liquidgas coexistence curve in a PT phase diagram end at the critical point?
-
An ideal gas is expanded reversibly and isothermally. Decide which of q, w, U, and H is positive, negative, or zero.
-
Your local newspaper started publishing puzzles of the following form: Parenthesize 6+0.6 to maximize the outcome. Wrong answer: 6+(0-6)=6+0= 6. Right answer: (6+0) 66.636. . Parenthesize 0.1.0.1+0.1...
-
Planck's radiation law tells us that the intensity of radiation per unit area and per unit wave- length A from a black body at temperature T is I(A) - 2hc21-5 ehc/AkBT -1 where h is Planck's...
-
Hack Wellington Co. (HWC) plans to take out a five-year term loan from a commercial bank for $2,500,000 at a stated interest rate of 6%. The contract of the term loan requires that the borrower makes...

Study smarter with the SolutionInn App


