Crystallographers have a rough rule of thumb that non-H atoms occupy around 18 3 in many
Question:
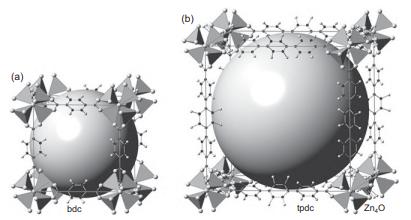
Crystallographers have a rough “rule of thumb” that non-H atoms occupy around 18 Å3 in many crystal structures. Based on this assumption, estimate the percent pore space in the tpdc MOF on the right of Figure 14.14.
Figure 14.14
Transcribed Image Text:
(a) bdc tpdc Zn0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The contents of the cell shown woul...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
After graduating from business school, George Clark went to work for a Big Six accounting firm in San Francisco. Because his hobby has always been wine making, when he had the opportunity a few years...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The Collectors Choice Inc. ( CCI), a luxury car import company, has an old warehouse at the Port of Miami, Florida, where it temporarily stores expensive sports cars and automotive parts that arrive...
-
For each polynomial function, complete the following in order. (a) Use Descartes rule of signs to determine the different possibilities for the numbers of positive, negative, and nonreal complex...
-
Find the wavelength of the K line in copper (Z = 29) if the wavelength of the K line in iron (Z = 26) is known to be equal to 193 pm.
-
A company issues $5,000,000, 7.8%, 20-year bonds to yield 8% on January 1, 2010. Interest is paid on June 30 and December 31. The proceeds from the bonds are $4,901,036. What is interest expense for...
-
Zeta Safety, Inc., began the year with $15,000 of common stock and $34,000 of retained earnings. On August 5, investors bought $19,000 of additional stock in the business. On October 22, the business...
-
The directors of Mylo Ltd. are currently considering two mutually exclusive investment projects. Both projects are concerned with the purchase of a new plant. The following data are available for...
-
Transform the given system into a single equation of second-order x = 5x1 + 2x2 x2 = -2x1 - 5x2. Then find x1 and x2 that also satisfy the initial conditions NOTE: Enter exact answers. x1(t) = x2(t)...
-
Consider the PtS net listed in Table 1.6. The dehydrated form of MOF-11 has composition Cu 2 (ATC) where ATC is 1,3,5,7-adamantane tetracarboxylate and contains a Cu paddle-wheel unit analogous to...
-
The first peak in the powder diffraction pattern of a mesoporous MCM material with 30 pores recorded with a wavelength of 1.54 is at 2.2 2. Estimate the thickness of the silica walls. A hexagonal...
-
Use rules of exponents to simplify the expression. Use positive exponents to write your answer. (3xy-3)-
-
PR/2 a+bsinx dx 7. Apply differentiation under integral sign to evaluate log absinx sinx 8. Define Gamma function. Apply Beta and Gamma function to evaluate: cos 60.sin* 30= 7 192 9. Find the area...
-
On April 1, 10,000 shares of $5 par common stock were issued at $22, and on April 7, 5,000 shares of $50 par preferred stock were issued at $104. Journalize the entries for April 1 and 7. If an...
-
i. A mobile base station (BS) in an urban environment has a power measurement of 45 W at 285 m. If the propagation follows an inverse 4th power law (Section 3.2.2), what is a reasonable power value,...
-
A professor once suggested the "Big Mac" index was a useful way to calculate the purchasing price parity of currencies around the world.He noticed the following: Cost of a Big Mac Hamburger (BMH)in...
-
Wells Fargo & Company, headquartered in San Francisco, is one of the nation's largest financial institutions. Suppose it reported the following selected accounts (in millions) as of December 31,...
-
Take a process that you encounter on a daily basis, such as the lunch cafeteria or the journey from your home to school/work, and identify the bottlenecks that limit the throughput of this process.
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
Give an example of two systems that are in equilibrium with respect to only one of two state variables.
-
Aerobic cells metabolize glucose in the respiratory system. This reaction proceeds according to the overall reaction 6O 2 (g) + C 6 H 12 O 6 (s) 6CO 2 (g) + 6H 2 O(l) Calculate the volume of oxygen...
-
Using acetylene as your only source of carbon atoms, outline a synthesis for 3-hexyne.
-
New customer service approaches should be communicated to all staff to ensure they provide consistent service that meets customer needs and expectations. Explain how managers can develop and...
-
The hormone glucagon is secreted by the pancreas in response to low blood sugar. Liver cells respond to glucagon by breaking down glycogen to form glucose, which is then release into the bloostream...
-
can tools like a swot analysis or other power-sharing management practices help alleviate the consolidation of power within organizations? How can we empower others rather than attempt to hold all...

Study smarter with the SolutionInn App


