State the KrgerVink notation for the predominant defects in each of the materials in Table 2.2. Table
Question:
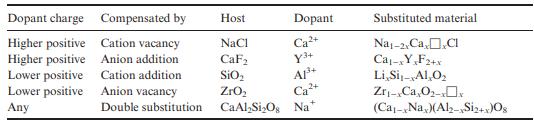
State the Kröger–Vink notation for the predominant defects in each of the materials in Table 2.2.
Table 2.2
Transcribed Image Text:
Dopant charge Compensated by Higher positive Higher positive Lower positive Lower positive Any Cation vacancy Anion addition Cation addition Anion vacancy Double substitution Host Dopant Ca+ ya+ NaCl CaF SIO ZrO CaAl-Si0 Na* C2 A1+ Substituted material Nat-2 Ca Cl Ca-xY,F2+x Li Si-AlO Zr-C0-xx (Ca-xNax)(Al2-Si2+x)08
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Na12CaCl VNa and Cana Ca...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
As an Econometrics student, you have just been hired by the new Zambian government to evaluate the relationship between monetary policy rate and economic growth. ( I ) Derive the OLS estimator for...
-
Consider the point P(-1,3,1). Use this point to answer questions 1 through 6 on this assignment. 1. Find an equation for a line containing P and passing through the origin.
-
After constructing a new manufacturing machine, five prototype integrated circuit chips are produced and it is found that two are defective (D) and three are acceptable (A). Assume that two of the...
-
I recently heard my neighbor discussing how one of our other neighbors lost his job. My neighbor assumed that the person who lost his job was probably lazy or not smart enough for the job, without...
-
The two-bar linkage shown is supported by a pin and bracket at B and a collar at D that slides freely on a vertical rod. Determine the force P required to maintain the equilibrium of the linkage. -S...
-
If a nations currency depreciates, are the reduced export prices that result unfair?
-
The shape of the temperature-entropy diagram for the Carnot cycle is a (a) Rectangle (b) Rhombus (c) Trapezoid (d) Circle.
-
Public Charity. The Kids Club of Clare County is a public charity under IRC Sec. 501 (c) (3). It had total support last year of the following: United Way support $80,000 Grant from dare County 70,000...
-
The atoms in a nickel crystal vibrate as harmonic oscillators with an angular frequency of 5.5x 1013 rad/s. The mass of a nickel atom is 9.75 x 10-26 kg. What is the difference in energy between...
-
Using the cell-parameter information, calculate the cell parameter expected for In 0.76 Ga 0.24 P 0.47 As 0.53 . Would this composition be lattice matched to any of the four possible end members:...
-
Suggest what type of twinning might occur in: (a) An orthorhombic structure with two cell edges approximately equal; (b) A structure with a monoclinc cell with = 90.1; (c) An orthorhombic structure...
-
Calculate the Van der Waals constants for carbon dioxide if its critical temperature Ter, = 304 K and critical pressure per = 73 atm.
-
Carbon monoxide (CO) contained in a piston-cylinder arrangement, initially at 5.3 bar and 497 K, undergoes an expansion to a final temperature of 224 K, during which the pressure-volume relationship...
-
II. A gas mixture, with the mole fraction listed below, enters the turbine of Figure at 1200kPa and 600K with an entering volume flow rate of 1.2m /s. The mixture leaves the turbine at 300K....
-
B-9-4 (To find the solution use m = 1 kg, m =1/4 kg, k =1 N/m,b=0 N /m, P=1, w=1 rad/s ). Hint: Transform the equations of motion from physical coordinates into modal coordinates to decouple the...
-
A 4x4 meter [45/-45]s balanced symmetric layup (note 4 plies) is used in a space applications where the change in temperature is AT=-100C. Once on orbit at this temperature, the sample experiences...
-
. Given G = ({A, S}, (a, b }, S, P) with P, defined by the production rules: S aAb | 2 AaAb | (i). Show that L(G)= {a"b": n0}. (ii). Show that G is equivalent to G where G= ({S}, {a,b), S, P) where P...
-
Guthridge Soap Corporation is evaluating a new soap cutting machine that could eliminate some direct labor costs. The machine would cost $900,000 per year and would cost $0.10 per bar to cut the...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Identify the reagent you would use to accomplish each of the following transformations: (a) Cyclobutanol bromocyclobutane (b) tert-Butanol tert-butyl chloride (c) Ethyl chloride ethanol
-
How many different alkenes will produce 2, 4-dimethylpentane upon hydrogenation? Draw them.
-
Using acetylene as your only source of carbon atoms, design a synthesis of pen-tanal. (Pentanal has an odd number of carbon atoms, while acetylene has an even number of carbon atoms): H.
-
Considering a common size statement for the financial statements here was made, what would be the the common size percentage for: Rent Expense Capital Cost of Goods Sold Creditors Control Gross...
-
Your client is closely following a publicly listed Australian company, however your client does not have the requisite skills to evaluate the company and, as such, has provided you with the following...
-
Place the following phasesa job moves through in order of processing: - 1. 2. 3. 4. 5. 6. Purge - 1. 2. 3. 4. 5. 6. Conversion - 1. 2. 3. 4. 5. 6. Input - 1. 2. 3. 4. 5. 6. Output - 1. 2. 3. 4. 5. 6....

Study smarter with the SolutionInn App


