Suggest oxidation states for the metal ions in each of the following materials: (a) TiS2, Lio.7 TiS2;
Question:
Suggest oxidation states for the metal ions in each of the following materials:
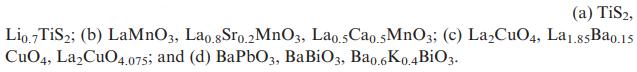
Transcribed Image Text:
(a) TiS2, Lio.7 TiS2; (b) LaMnO3, Lao.8Sro.2 MnO3, Lao.5Cao.5MnO3; (c) LaCuO4, La1.85Ba0.15 CuO4, LaCuO4.075; and (d) BaPbO3, BaBiO3, Ba0.6K0.4BiO3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Oxidation states a Ti4 note difference from FeS2 in previous Problem Li Ti ...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Suggest oxidation states for the metal ions in each of the following materials: (a) FeO, Feo.8720, Fe3O4, FeS2; (b) FeTe, Fe1 Te; (c) LaOFeAs, LaO0.9Fo.1 FeAs; and (d) YBaFeO5, NdBaFeO5.5, and...
-
What strategy would be the most useful to companies interested in Brazilian investment?
-
State the predicted ionic charge of metal ions in each of the following groups of elements. (a) Group IA/1 (b) Group IIA/2 (c) Group IIIA/13 (d) Group IVA/14.
-
a. Distinguish between internal common equity and new common stock. b. Why is there a cost associated with internal common equity? c. Describe two approaches that could be used in computing the cost...
-
Derive an expression for the magnitude of the couple M required to maintain the equilibrium of the linkage shown.
-
1. Would you suggest that Hotel Paris implement a competency based pay plan for its non-managerial staff? Why or why not? Outline what they need to do. 2. Devise a ranking job evaluation system for...
-
The Mollier diagram is a plot of (a) Temperature versus enthalpy (b) Temperature versus entropy (c) Enthalpy versus entropy (d) Temperature versus internal energy.
-
Income statements for Gaidar Company for 2014 and 2015 follow. Required Round all percentages to one decimal point. a. Perform a horizontal analysis, showing the percentage change in each income...
-
Social Media Audit- Anthropologie For each platform: Instagram, Facebook, Pinterest, Twitter, and TikTok How are they using each platform? How are they creating engagement? What are their engagement...
-
A brown sample of zinc oxide was found to have the hexagonal wurtzite structure with a = b = 3.2495 , c = 5.2069 ( = = 90; = 120). Chemical analysis gave 80.765% Zn by mass. Density measurements...
-
TiO with a 1:1 ratio of Ti:O was synthesized and found to have a NaCl-related structure with a = 4.1831 and an experimental density of 4927 kg/m 3 . Comment on these values.
-
Give as many examples of cultural differences as you can, using information from this text and your own experiences.
-
7) A rigid bar is supported by three rods in same vertical plane and equi distant. The outer rods are of brass and of length 600mm and diameter 30mm. The central rod is of steel of 900 mm length and...
-
3 A block of mass M-25 kg is attached to the cord wound on the drum of diameter 100 mm and it is allowed to fall under the action of gravity (see Figure B3). The drum is firmly attached to gear A,...
-
(b) The following C++ function multiplies natural numbers x and y by repeated addition. Derive, and prove correct, the number of "+" operations this method executes. int multiply (int x, int y) ( }...
-
4. 5. A drug is to be sold in gelatin capsules, which have an approximately cylindrical shape. To hold the required dosage, the capsules must have a volume of 120 cubic millimeters. What are the...
-
The belt in the given image moves at a steady velocity Vand skims the top of a tank of oil of viscosity J. + Of Oil, depth h Moving belt, width b What power P in watts is required if the belt moves...
-
Calista Company manufactures electronic equipment In 2009, it purchased the special switches used in each of its products from an outside supplier. The supplier charged Calista $2 per switch....
-
Juanita owns a home in Richardson, TX. She purchases a Homeowners Policy (HO-3) from Farm State Ins. Co. The policy provides $100,000 in liability coverage (coverage E) and $5,000 in Med Pay coverage...
-
Propose a mechanism for each of the following reactions: a. b. c. d. ,* ,*
-
Identify reagents that can be used to achieve each of the following transformations: a. b. c. d. e. f. Br Br HO,
-
Draw a Lewis structure for each of the following ions; in each case, indicate which atom possesses the formal charge: a. BH 4 b. NH 2 c. C 2 H 5 +
-
What are some of the factors that should be taken into account when you are recruiting and selecting?
-
A person donates a plot of land within a developed residential area to an entity. The land is currently used as a playground. The donor specifies that the entity must continue using the land as a...
-
CPA Exam Adapted: A company purchased a computer on May 1, for $12,000 with an estimated salvage value of $1,500 and a 3-year life. What is the depreciation expense for the year ended December 31,...

Study smarter with the SolutionInn App


