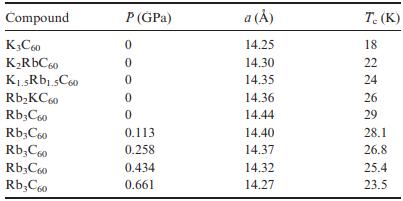
The table below contains data for the alkali-metal fullerides A 3 C 60 . (a) Give a
Question:
The table below contains data for the alkali-metal fullerides A3C60.
(a) Give a brief explanation for the Tc changes observed in the 0 GPa data.
(b) Give a brief explanation for the pressure dependence of Tc in Rb3C60.
(c) Comment on differences in Tc between the two series.
Transcribed Image Text:
Compound K3C60 KRbC60 K.5Rb1.5C60 RbKC60 Rb3C60 Rb3C60 RbC60 Rb3C60 RbC60 P (GPa) 0 0 0 0 0 0.113 0.258 0.434 0.661 a () 14.25 14.30 14.35 14.36 14.44 14.40 14.37 14.32 14.27 T. (K) 18 22 24 26 29 28.1 26.8 25.4 23.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
abc The data relate to Figure 1210 where the dependence of T c on the volume per C 60 was presented ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
You are currently willing to pay $25,000 to purchase a perpetuity which will pay you and your heirs $2,250 each year forever (the first $2,250 payment will be received one year from today). If your...
-
Watt Wholesale Company purchases merchandise from a variety of manufacturers and sells the merchandise to a variety of retailers. All sales are subject to a cash discount (2/10, n/30). Watt uses a...
-
Discuss whether the following supply of goods and services are taxable events in GST: 1 . A company having warehouse in Mumbai shifted some raw material to its factory in Baroda 2 . An Indian company...
-
Julio sold his corporation to a competitor, Exeter LLC, for $100,000,000. Julio incorporated his business 17 years ago by investing $500,000 plus his proprietary know-how. There have been no other...
-
Find the threshold energy of gamma quantum required to form (a) An electron-positron pair in the field of a stationary electron; (b) A pair of pions of opposite signs in the field of a stationary...
-
As you now know, the jury decides the facts of the case, and the judge decides matters of law. The judge instructs the jury on the law. But did you know that the judge does NOT instruct the jury on...
-
The charter of Maple Leaf Capital Corporation authorizes the issuance of 1,000 shares of preferred stock and 10,000 shares of common stock. During a two-month period, Maple Leaf completed these...
-
Abe Technologies provides maintenance service for computers and office equipment for companies throughout the northeast the sales manager is elated because she closed a $300,000, 3-year maintenance...
-
(3 marks) Question 2 (32 marks) Jenny has $200,000 to invest and is considering the merits of two securities. He is interested in the common shares of X Co. and Y Inc. The possible monthly rate of...
-
Rb x Cs 3x C 60 compounds have T c = 26.9 K, 32.9 K, and 31.8 K and room-temperature cell parameters a = 14.70 , 14.60 , and 14.45 for x = 0.35, 1.0, and 2.0,respectively. Predict the dependence of...
-
Draw a sketch of the fcc structure of C 60 . Show the positions of octahedral and tetrahedral holes and state the number of each. Assuming that C 60 has a radius of 5 , estimate the cubic cell...
-
For each of the following fund types, indicate its fund category by placing either GF for governmental funds, PF for proprietary funds, or FF for fiduciary funds in the space provided before each...
-
LeAnne is exhausted after another long day of answering the phones at the law firm of Boardman, Holloway, and Shriver. She says those three names several hundred times a day, followed by "How my...
-
Suppose that instead of wealth, the consumer starts with a bundle of goods z (not necessarily her optimal bundle), and can buy and sell goods at prices p. Suppose that all the goods are regular....
-
Errors in trial balance Identify the errors in the following trial balance. All accounts have normal balances. Ensemble Co. Unadjusted Trial Balance For the Year Ending December 31, 20Y8 Debit Credit...
-
Multiply. 4 (x3 + x+x+1)(13x-2)
-
How can I create a map of Berlin with Rstudio? I would also like to create a linear regression with the NAO values and precipitation values. The precipitation values are monthly. Therefore, all NAO...
-
Looking back with perfect hindsight, what should the brand manager for Nestls Kit Kat have done when the Greenpeace e-mails first appeared?
-
Anne is employed by Bradley Contracting Company. Bradley has a $1.3 million contract to build a small group of outbuildings in a national park. Anne alleges that Bradley Contracting has discriminated...
-
Calculate the pressure exerted by Ar for a molar volume of 1.31 L mol 1 at 426 K using the van der Waals equation of state. The van der Waals parameters a and b for Ar are 1.355 bar dm 6 mol 2 and...
-
A van der Waals gas undergoes an isothermal reversible expansion under conditions such that z > 1. Is the work done more or less than if the gas followed the ideal gas law?
-
When isopropylbenzene (cumene) is treated with NBS and irradiated with UV light, only one product is obtained. Propose a mechanism, and explain why only one product is formed. Br NBS hv
-
1. Use the phase diagram from the lecture notes on the water sugar system. You start off with a 100 g mixture of 70 wt% sugar and 30 wt% water, heat above the solubility limit to dissolve the sugar,...
-
2. (10 pts.) Identify the point symmetry elements of the structures for which the given directions are equivalent. Enumerate the elements (i.e., the individual symmetry operations) that make up the...
-
1. (10 pts.) In metals the mechanical properties are often determined by the motion of dislocations. Dislocations are fundamentally a crystalline defect in materials. a. (5 pts.) In the FCC structure...

Study smarter with the SolutionInn App


