Using the cell parameters quoted, confirm the form of Equation (2.9). Equation (2.9) X= y 2.21 -0.053y
Question:
Using the cell parameters quoted, confirm the form of Equation (2.9).
Equation (2.9)
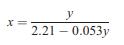
Transcribed Image Text:
X= y 2.21 -0.053y
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Using ax y a InP and introducing approp...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Theresa Pratt works in the business office at Mills College, a technical college in Council Bluffs, Iowa. She has created a workbook to store and analyze registration and employee data. The workbook...
-
Nadia Khalif is the office manager for the Conyers Law Offices in Dallas, Texas. The firm is planning to move its computer network into the cloud so that everyone working on a case can access...
-
Determine the depreciation for the year of acquisition and for the following year of a fixed asset acquired on October1for $500,000with an estimated life of five years, and residual value of...
-
Suppose Nordic Boots was making production decisions for the following six boots: ot Price E(Demand) Std. Dev (Demand) Co Cu Exp. Profit Max. Profit A 220 1000 400 35.2 52.8 1101 39201 52800 230 2000...
-
The mechanism shown is acted upon by the force P. Derive an expression for the magnitude of the force Q required for equilibrium. OE
-
Many individuals prefer to have insurance (health insurance, car insurance, etc.) rather than not, even if the expected value of their wealth is higher without insurance. What does this imply about...
-
Consider the following cash flow profile, and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically...
-
(Pension Expense, Journal Entries for 2 Years) Gordon Company sponsors a defined benefit pension plan. The following information related to the pension plan is available for 2010 and 2011. (a)...
-
Why does flexibility in NoSql make it easier? What is an example.
-
GaAs 1x P x has a unit-cell parameter of 5.59 . Calculate x and estimate the band gap(E g ) of the material, given aGaAs = 5.65 , Eg = 1.42 eV; aGap = 5.45 , E = 2.24 eV.
-
Using the cell-parameter information, calculate the cell parameter expected for In 0.76 Ga 0.24 P 0.47 As 0.53 . Would this composition be lattice matched to any of the four possible end members:...
-
How do you decide whether to give the negative directly or to buffer it?
-
Consider the following method which searches a linked list of integers for a value. static boolean checkList (Node node, int target) { } if (node == null) return false; if (node.data == target)...
-
Okra, Inc. is a young start-up company. No dividends will be paid on the stock initially, because the firm needs to plow back its earnings (i.e., not to pay out dividends) to fuel growth. Five years...
-
You are the room leader in the toddlers' room in an Early Childhood Education and Care Service. By working together with other educators, for the team create a nurturing and positive environment for...
-
Suppose you just won the state lottery, and you have a choice between receiving $2,700,000 today or a 20-year annuity of $250,000, with the first payment coming one year from today. Assuming both...
-
Project Management Openings This case will address the changing human capital landscape in the industry. State of the Industry Deliver a comprehensive analysis of staffing concerns in the project...
-
This chapter identified many decision contexts in which variable costing information is more relevant than absorption costing. However, absorption costing is still used by many companies and remains...
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
Consider the structures of cis-1,2-dimethylcyclopropane and trans-1,2-dimethylcyclopropane: (a) Which compound would you expect to be more stable? Explain your choice. (b) Predict the difference in...
-
Draw bond-line structures using wedges and dashes for the following compounds: a. b. c. d. e. CH, - - - - CH- Et - - . - Me
-
The following four reactions will be the focus of the upcoming chapters (substitution and elimination reactions). Draw the curved arrows that accomplish each of the transformations shown: a. b. c. d....
-
Explain, Free riding, where individuals benefit from public goods without contributing, is a significant issue, especially in larger groups, posing a dilemma in achieving optimal provision. Public...
-
Dillo Mfg. builds an average of 20 compressors weekly, with standard deviation of 2 (assume normal distribution). Each compressor requires a drive belt. Drive belt inventory levels are reviewed every...
-
Give a school-based example of two of the following: positive reinforcement, negative reinforcement, punishment, or extinction. Discuss at least two benefits of conducting an FBA to address problem...

Study smarter with the SolutionInn App


