Write out half equations, the cell reaction, and determine the standard potential of the following cells given
Question:
Write out half equations, the cell reaction, and determine the standard potential of the following cells given standard electrode potentials
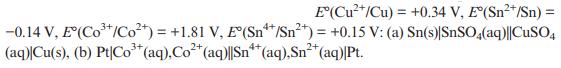
Transcribed Image Text:
E(Cu+/Cu) = +0.34 V, E(Sn*/Sn) = -0.14 V, E(Co*/Co) = +1.81 V, E(Sn/Sn+) = +0.15 V: (a) Sn(s)|SnSO4(aq)||CuSO4 3+ 2+ (aq)|Cu(s), (b) Pt|Co+ (aq), Co+ (aq)|Sn** (aq), Sn+ (aq)|Pt.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Cu 2 2e Cu E 034 V higher standard potential defines the oxidizing agent Sn 2 2e ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
As in Example 6L.1, you are planning to use a Daniell cell to power a model electric car. However, you find that you do not have standard solutions available. You have only dilute solutions, and you...
-
Write the cell reaction and electrode half-reactions and calculate the standard emf of each the following cells: (a) Ptl C12 (g) I HCl (aq) 11 K, Cr04 (aq) IAg, Cr04(s) IAg (b) Pt 1 Fe3+(aq),Fe2+(aq)...
-
The list below gives the standard electrode potentials for five half-reactions. Cu 2+ (aq) + e Cu(s) E = +0.34 V Fe 2+ (aq) + 2e Fe(s) E = 0.44 V Fe 3+ (aq) + e Fe2+(aq) E = +0.77 V I 2 (aq)...
-
Blackmon Company provides locator services to the city transportation departments. Blackmons service involves installing a dedicated hardware transmitter in each city bus. This transmitter provides...
-
Determine the wavelengths of spectral lines appearing on transition of excited Li atoms from the state 3S down to the ground state 2S. The Rydberg corrections for the S and P terms are 0.41 and ...
-
Al a Mode, Inc., is considering one of two investment options. Option 1 is a $60,000 investment in new blending equipment that is expected to produce equal annual cash flows of $16,000 for each of...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically attractive?...
-
Use the following information to prepare a classified balance sheet for Alpha Co. at the end of 2016. Accounts receivable .....$26,500 Accounts payable ....... 12,200 Cash ............ 20,500 Common...
-
Pawan is pursuing MBA and shares a room with you in the private hostel. He desperately needs a job after completing his course because of educational loan and weak economic background. In his last...
-
In its charged state, a leadacid battery contains PbO 2 and Pb electrodes. The half equations that occur during discharge can be written as below. Give the overall cell equation and estimate E cell....
-
What is unusual about the superconducting phases Nd 2x Ce x CuO 4 and Sr 1x Nd x CuO 2 ?
-
Use recursion to write a C++ function for determining if a string s has more vowels than consonants.
-
Provide your perspective of common law and the development of rules regarding the rights and liabilities of innkeepers under the common law system. Provide at least one example of common law related...
-
Equity law came as a result of shortcomings of common law.What was the common law based on?
-
All of the following are true of ESG investing EXCEPT _____. Only uneducated investors tend to use it. The investors in ESG investing consider the impact of the investment on several stakeholders. It...
-
A company's 5-year bonds are yielding 7.75% per year. The real risk-free rate (r*) is 2.3%. The average inflation premium is 2.5%; and the maturity risk premium is estimated to be 0.1 (t - 1)%,...
-
what are the metro (grocery store)'s strengths in its reward system? B) Based on the opportunities below, what are the threats to those changes? Metro app upgrades Option to receive update emails on...
-
Juan Sanchez writes the following note on the back of an envelope: I, Juan Sanchez, promise to pay Kathy Martin or bearer $500 on demand. Is this a negotiable instrument? Discuss fully.
-
What types of questions can be answered by analyzing financial statements?
-
Compound A has molecular formula C 5 H 11 Br. When compound A is treated with bromine in the presence of UV light, the major product is 2,2-dibromopentane. Treatmentof compound A with NaSH (a strong...
-
In calculating H o R at 285.15 K, only the H o f of the compounds that take part in the reactions listed in Tables 4.1 and 4.2 (Appendix B, Data Tables) are needed. Is this statement also true if you...
-
Predict the products when each of the following compounds is treated with NBS and irradiated with UV light: (a) (b) (c) (d)
-
How does lean production improve lead times and customer responsiveness?
-
Could you please briefly explain optimized packaging and its application viability in developing and under developed countries?
-
The provider orders Pronestyl . Medication label: Pronestyl procainamide injection . How much medication should be administered? Drug category: The provider orders Compazine 7 mg IM. Medication...

Study smarter with the SolutionInn App


