Fill in the missing steps from (4.9.30) to (4.9.31). First, write a Sommerfeld expansion to second order
Question:
Fill in the missing steps from (4.9.30) to (4.9.31). First, write a Sommerfeld expansion to second order in T for the total electron number N. The lowest-order terms of both U and N can then be expanded as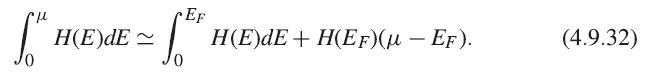
Using the constraint that N is constant for all temperatures, you can obtain a relation for μ in terms of T and substitute this into the equation for U, and then take its derivative, keeping terms to lowest order in T.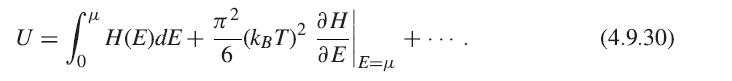
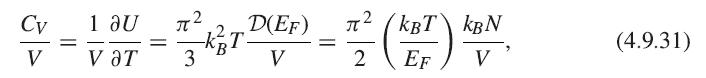
Transcribed Image Text:
[th B EF H(E)DE ~ ~ f H(E)dE+ H(EF)(µ – Ef). (4.9.32)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
The number of electrons is equal to N NEDE dE 4919 Following the same logic as equations 49254930 ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following selected financial information is available for Best, Inc. Amounts are in millions of dollars. Required a. Divide the class into groups of four or five students each. Organize the...
-
The following selected financial information is available for ROC, Inc. Amounts are in millions of dollars. a. Divide the class into groups of four or five students each. Organize the groups into...
-
O O Multiple Choice Which of the following sequence of events best describes an online intrusion? A. Malware enters the device, then opens a backdoor, leading to an open communications link on the...
-
XYZ Co. It is currently trading at $5 per share and has announced a $0.50 per share dividend payable next year. Using historical information, one analyst estimates XYZ Co.'s dividend growth rate is...
-
The following information is available for the pension plan of Talkspace Corporation for the year 2014. Actual and expected return on plan assets ....... $ 65,000 Benefits paid to retirees...
-
Two investments generated the following annual returns: a. What is the average annual return on each investment? b. What is the standard deviation of the return on investments X and Y? c. Based on...
-
Choose an appropriate closed system and draw a bar diagram representing the energy conversions and transfers that occur during each process of Checkpoint 7.9: (a) a ball launching as the compressed...
-
Order Up, Inc., provides order fulfillment services for dot.com merchants. The company maintains warehouses that stock items carried by its dot.com clients. When a client receives an order from a...
-
(a) NP and Co. has imported goods for US $ 7,00,000. The amount is payable after three months. The company has also exported goods for US $ 4,50,000 and this amount is receivable in two months. For...
-
Show that (4.8.21) satisfies the detailed balance condition (4.8.20). A critical step in the proof is to invoke energy conservation, E k + E k1 = E k2 + E k3 . f(Ek3)f(Ek)(1(Ek ))(1 (Ek)) (Ek) (Ek...
-
Write down the quantum Boltzmann equation for the evolution of a population of fermionic particles interacting with a population of phonons through an interaction term of the form where a k3 is the...
-
The concentrations of magnesium, calcium, and nickel(II) ions in an aqueous solution are 0.0010 mol L 1 . (a) In what order do they precipitate when solid KOH is added? (b) Determine the pH at which...
-
hat methodologies and metrics are employed in quantifying biodiversity, considering taxonomic, genetic, and functional dimensions, and how do these measurements inform conservation strategies?
-
Moana and Jane are employed by Sobecal (a pharmaceutical company) to research and develop new pharmaceuticals. Moana and Jane are full of enthusiasm for research, so at weekends they often carry out...
-
Why is having knowledge and the capability to creatively apply that knowledge in creating unique solutions for their buyers so important for today's salesperson?
-
In the event that oil prices increase sharply, or there is a Global spike in terrorist attacks: Does the event affect aggregate demand (AD) or aggregate supply (AS)? Is the event's effect on AD or AS...
-
Explain, The experimental studies in Section 5 challenge the idea of self-interest in economics and government policies. They show that people are motivated by factors beyond material incentives,...
-
The MaxwellBoltzmann distribution function f(u) increases at small values of u and decreases at large values of u. Identify the parts of the function responsible for this behavior.
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
Draw the major product(s) expected when each of the following alkynes is treated with aqueous acid in the presence of mercuric sulfate (HgSO 4 ): (a) (b) (c) (d) (e)
-
Identify the alkyne you would use to prepare each of the following ketones via acid-catalyzed hydration: (a) (b) (c)
-
The amino acid glycine dimerizes to form the dipeptide glycylglycine according to the reaction 2Glycine(s): Glycylglycine(s) + H 2 O(l) Calculate ÎS, ÎS surr , and ÎS suniverse at T...
-
the tuition fee for maths tuition for john is expected to be 6500 per year if he attend tuition for 4 year starting from 4 years from now how much does he have to have in her account which yields 6...
-
Pioneer Inc. wants to invest $557,302 today. The expected returns in years 1, 2, and 3 are $247,615, $180,383, and $335,481, respectively. If the rate of return on investment must be at least 14%,...
-
A 25-year-old Latin, G1P1, cisfemale presents to the office with a 6-month history of amenorrhea; reports having regular menstrual cycles every month up until about 1 year ago when her menstrual...

Study smarter with the SolutionInn App


